Getting a leg up on chronic exertional compartment syndrome in the deep compartment
Alicia Filley assesses lower leg pain caused by chronic exertional compartment syndrome of the deep posterior compartment.

Lower leg pain is a common complaint among runners. Pain deep within the calf that starts after 20 to 30 minutes of exercise and resolves with rest is likely caused by chronic exertional compartment syndrome (CECS) of the deep posterior compartment. The pain may be described as a burning, aching, bursting, or tightness along the posterior medial border of the tibia. Pain or numbness may extend to the medial aspect of the foot.
The pain consistently manifests itself shortly after starting an activity and continues to worsen until the athlete is forced to stop the activity. With chronic deep posterior compartment syndrome (CDPCS), the muscles of the calf may feel swollen or tense upon physical exam, especially immediately after exercise. Pain may be present upon aggressive palpation or passive ankle dorsiflexion. Pain, numbness along the posterior-medial aspect of the calf, and weakness in toe flexion, ankle inversion and plantar flexion, may continue for some time after exercise, but usually resolve with a day’s rest, only to re-appear when the athlete returns to training. The pain occurs bilaterally in 80-95% of athletes with CECS, and involves the deep posterior compartment in 32%-60% of all cases of CECS(1).
Anatomy
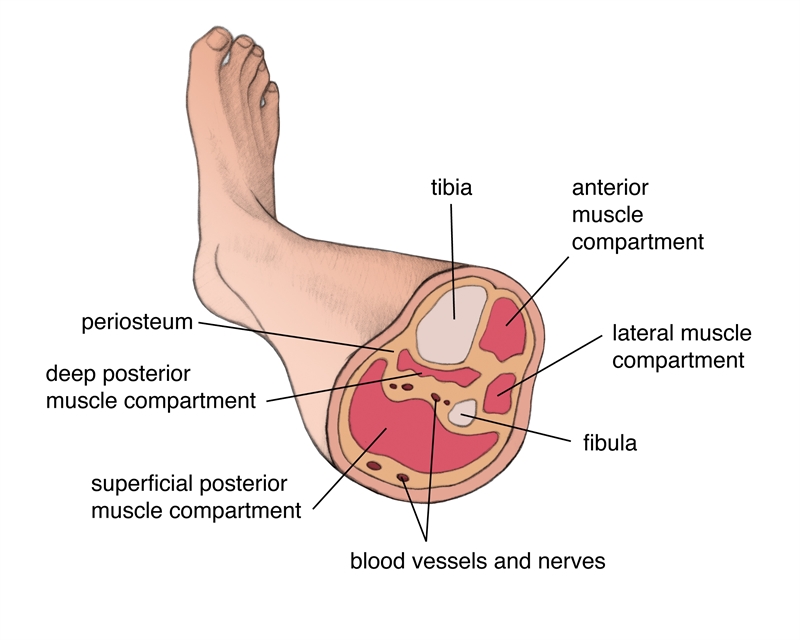
There are four fascial compartments within the lower leg: anterior, lateral, superficial posterior, and deep posterior (see figure 1). A compartment consists of a fascial sheath and the contents within – muscles, nerves, and blood vessels. Some consider the tibialis posterior to be a compartment unto itself because of its own fascial covering, but for practical purposes, it is included in the deep posterior compartment. Chronic exertional compartment syndrome most often affects the anterior compartment, followed by the deep posterior compartment in frequency(1).
Within the deep posterior compartment lie the tibialis posterior, flexor digitorum longus, flexor hallucis longus, and popliteus muscles. The posterior tibial nerve, artery, and vein also course through the deep posterior compartment. The muscles of the posterior compartment assist with inversion and plantar flexion of the foot and the posterior tibial nerve (L5-S1) innervates them.
Figure 1: Muscular compartments of the lower leg
Under pressure
Compartment syndrome occurs when the pressure of the fluid inside the compartment becomes so great that it restricts blood flow into the muscles within the compartment. Acute compartment syndrome, caused by extreme swelling after a traumatic event, is a medical emergency and requires immediate treatment with a fasciotomy before the lack of blood flow results in tissue death. Chronic exertional compartment syndrome also occurs due to the build-up of pressure within the compartment, but differs from the acute process in that the increased pressure occurs with exercise, and resolves when the activity is stopped.
During strenuous exercise, muscle volume normally increases up to 20%(2). If the muscle is hypertrophied or the fascia is less compliant, there is less room within the compartment for expansion and the pressure within the compartment increases. It is assumed that, as in acute compartment syndrome, the flow of oxygenated blood into the muscles is impeded if the pressure within the muscle is greater than the fluid pressure within the vessels themselves. To measure this pressure, clinicians use a needle catheter inserted into the compartment. The pressure is generally measured at rest, one minute after exercise, and five minutes post-exercise.
The only way to accurately diagnose CECS is by measuring the pressure within the compartment. This is done using a needle catheter inserted along the undersurface of the medial tibial border. Measurements of more than 15 mmHg at rest, 30 mmHg one minute after exercise, or 20 mmHg at five minutes post-exercise signify CECS(2).
Low flow
Theoretically, the pain with CECS is caused by the resulting ischemia, or cell death, within the muscles and nerves when blood flow is compromised. Studies attempting to confirm this theory fail to demonstrate the ischemic changes consistent with such levels of pain, except under extreme tissue pressure (≥ 160mmHg)(2). Calling the ischemia theory into question, researchers in Victoria, Australia, examined 34 patients under thallium-201 single-photon emission tomography(3). This imaging measured the perfusion within the muscles of the offending compartments. Twenty-five of the patients in the study had CECS confirmed by elevated compartment pressure, while nine had normal pressure but positive leg pain and served as controls. The researchers found no significant difference between the perfusion of those with CECS and those without, suggesting there is another etiology for the pain experienced.
Alternative theory
Normal fascial tissue is a thick connective tissue with little elasticity. Some theorize that in individuals with CECS, the fascia surrounding the compartment is less compliant than in others, due to repetitive loading on the fascia and its attachments to the bone1. To discover what type of histological changes may occur within the fascia in these individuals, researchers at the University of Melbourne explored the cellular nature of the fascia in individuals with CDPCS(1). In this case-controlled study, ten males and nine females with CDPCS underwent fasciotomy after conservative measures (not defined within the study) failed to improve the condition. Tissue samples were taken from the fascia of the deep posterior compartment and compared with control tissue samples taken from eleven autopsy subjects.
Interestingly, while the tissue samples of each of the subjects with CDPCS differed from one another, they did not differ significantly from the controls in measures of fibrocytic activity, chronic inflammatory cells , or vascular proliferation as assumed. However, the subjects differed significantly from controls in the degree of collagen organization. The researchers were surprised to find that the collagen in the fascia of the subjects showed more regular organization than that of the controls. While the exercise history of the cadaver subjects was not available, it was assumed that their collagen would be more organized. Instead, the alignment in the cadaver fascia was quite irregular.
Researchers hypothesized that the regularity found in the collagen arrangement in subjects with CDPCS was due to the chronic remodeling process experienced as a result of continuous strain on the fascia. This study was small, measured only a few variables, and the controls were not equally matched for activity with the subjects. However, noting changes within the fascia warrants further exploration to determine if the pain from CDPCS occurs due to the stimulation of the pain receptors within the fascia or the periosteum where it attaches to the bone, rather than ischemia.
Treatment options
It’s possible, then, that repeated strain and remodeling of the fascia, as hypothesized by the researchers at the University of Melbourne, decrease the pliability of the fascia(1). Therefore, stopping the repeated strain and increasing fascial compliance would seem to be the targets for treatment. Biomechanical analysis may reveal longstanding patterns of movement that have added stress to the calf over a prolonged period of time.
Exploring this premise, researchers in West Point, NY, evaluated the running technique of ten armed servicemen with a diagnosis of CECS of the anterior compartment(4). Each of these patients was awaiting fasciotomy for the treatment of the CECS. Each underwent a six-week program of running re-training to learn a forefoot-strike running technique. The program consisted of running analysis, drills, exercises, and feedback. After six weeks, post-run anterior compartment pressures and reported pain scores decreased significantly, while running distances increased significantly in all subjects. Positive results continued for one year after intervention in all ten subjects.
This study was limited in size, analyzed patients with anterior compartment CECS, not CDPCS, and did not include subject controls with whom to compare results. However, all subjects avoided surgery with a six-week intervention program of three 45-minute sessions per week. This speaks dramatically to the need for biomechanical analysis of all athletes with CDPCS. If the cause of strain can be eliminated, the hypothesis is that the fascia can heal and function normally again.
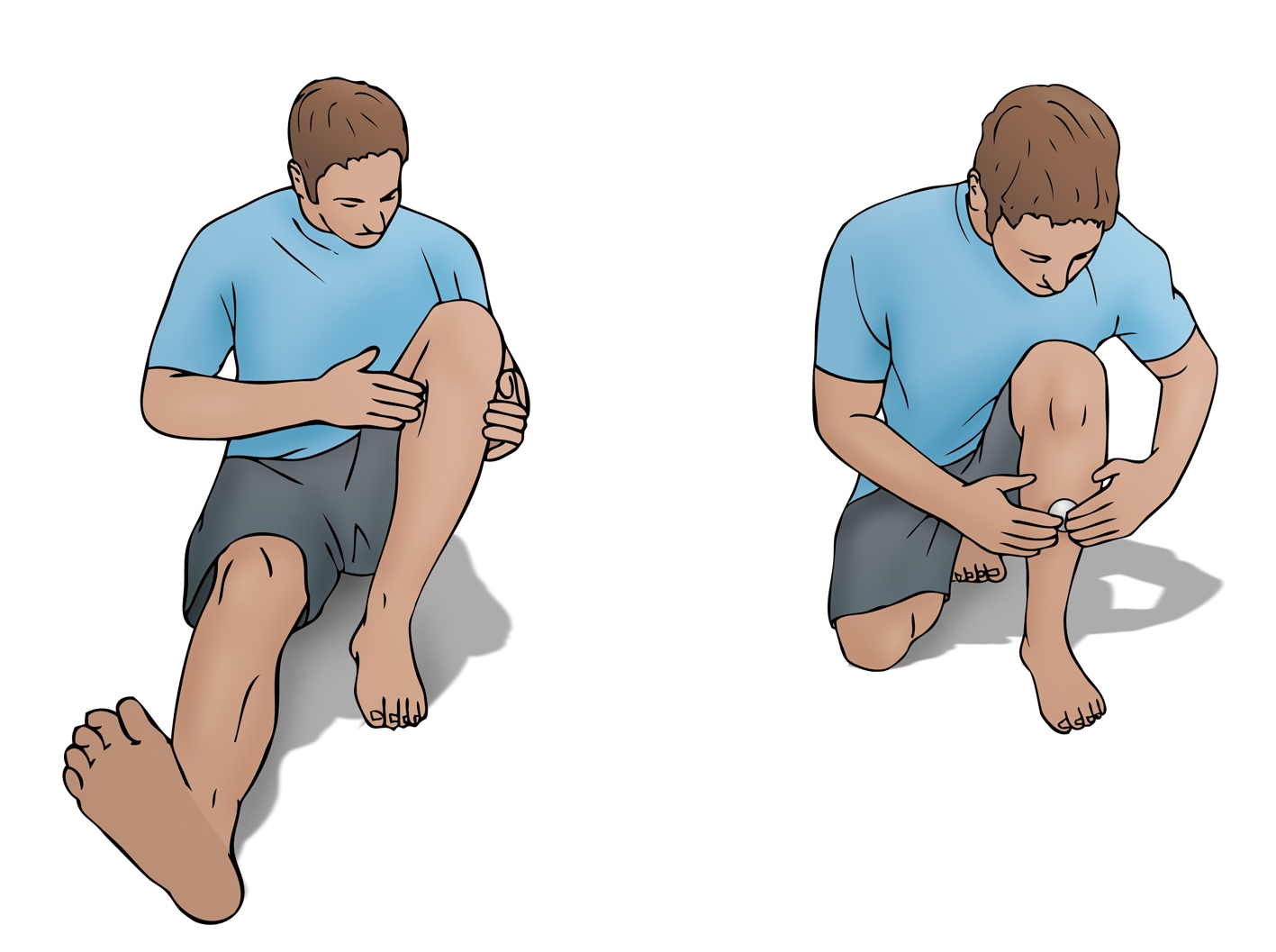
The biomechanical analysis should include an evaluation of strength, range of motion, skeletal alignment, and equipment, including shoes, orthotics, and running surfaces. Fascial compliance and nerve mobility can be evaluated and treated with stretches and exercises (see figures 2,3 and 4). Training schedules should also be assessed as year-round sport eliminates the opportunity for off-season cross-training and a break from continuous strain.
Figure 2: Stretch for the deep posterior compartment
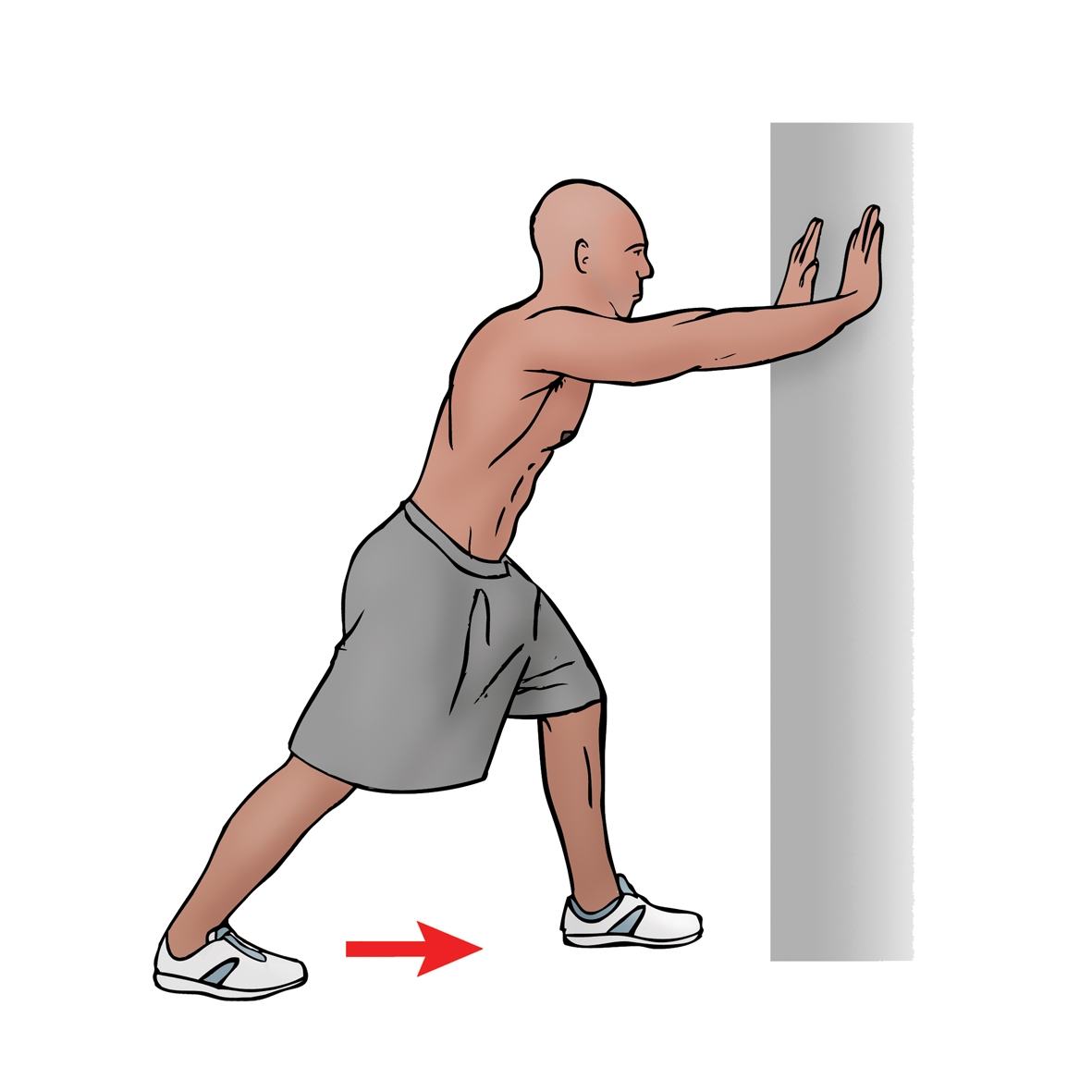
Figure 3: Neural glide for the tibial nerve
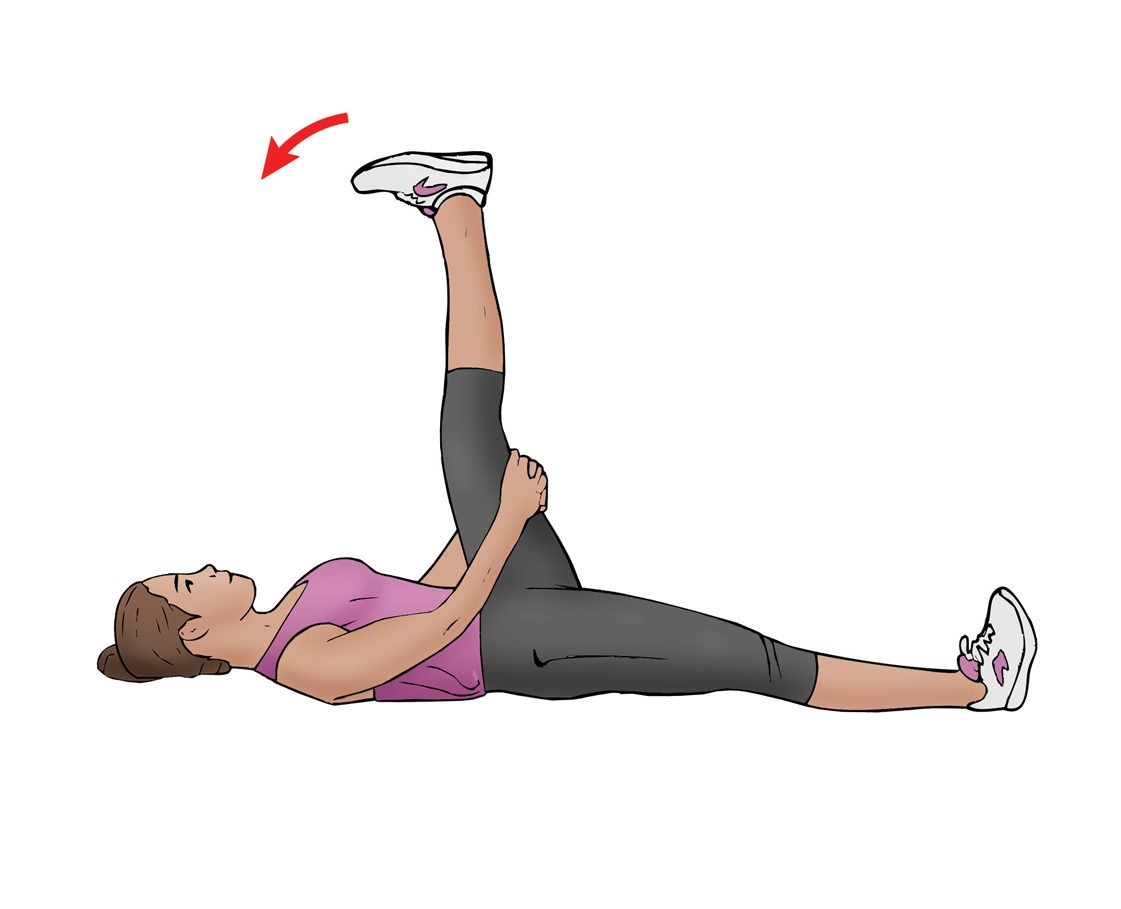
Figure 4: Mobilization of the fascia of the deep posterior compartment
Easing the pressure
In acute compartment syndrome, the way to relieve the build-up of pressure and save the tissue within the compartment from ischemia is to release the fascia through a fasciotomy. The assumption for the surgical management of CECS is the same, that releasing the fascia will normalize the perfusion of the compartment and relieve the pain. Current research, showing perfusion deficits do not exist within the compartment, questions the perfusion theory of pain, and thus the benefit of fasciotomy.
A researcher at the University of California reviewed the literature on the effectiveness of fasciotomy for CDPCS(5). Her review revealed that the often quoted 80% success rate of fasciotomy treatment for CECS only included rates for treatment of the anterior compartment, not the deep posterior compartment. She proposed, therefore, that a difference exists between the success of anterior and deep posterior compartment fasciotomy, as determined by patient satisfaction.
Seven studies met criteria for inclusion in the meta-analysis. The review revealed that there was a significant difference between the outcome satisfaction of those with anterior compartment fasciotomy (83%) and those with deep posterior compartment fasciotomy (56%). Closer scrutiny revealed studies with patient satisfaction levels at 75%-100% satisfied reported return to activity levels of only 50%-75%(5). In addition, the incidence of complication from the procedures in the studies reviewed ranged from 4% to 90% of cases.
There are several factors that may contribute to the low satisfaction scores. Since decreased perfusion is likely not the cause of pain in CDPCS, a fasciotomy may not address the problem at all. Accessing the deep posterior compartment is difficult, thus releasing the fascia along the entire compartment is challenging. Surgeons may not truly release the entire compartment in all cases. Rehabilitation standards also differ among practitioners, thus adding incomplete rehabilitation as a possible contributor to poor satisfaction outcomes.
Conclusion
Surveys show that 26%-33% of athletes with exercise-induced lower leg pain have CECS in one of the compartments of the lower leg(1). Pain on exertion in the calf of the lower leg is often caused by CDPCS. The prior assumption that the onset of pain after 20-30 minutes of exercise is due to increased pressure and decreased blood flow within the compartment is now under scrutiny. More likely, fascial strain is the cause of pain and the resulting decreased compliance is what causes the increased compartment pressure(2). To improve outcomes using conservative treatment, follow a systematic rehabilitation program, with emphasis on biomechanical analysis and correcting the factors that may have instigated the problem in the first place, namely, biomechanical deficits in running technique, fascial and nerve immobility, decreased range of motion, and muscle weakness.
You need to be logged in to continue reading.
Please register for limited access or take a 30-day risk-free trial of Sports Injury Bulletin to experience the full benefits of a subscription. TAKE A RISK-FREE TRIAL
TAKE A RISK-FREE TRIAL
Newsletter Sign Up
Subscriber Testimonials
Dr. Alexandra Fandetti-Robin, Back & Body Chiropractic
Elspeth Cowell MSCh DpodM SRCh HCPC reg
William Hunter, Nuffield Health
Newsletter Sign Up
Coaches Testimonials
Dr. Alexandra Fandetti-Robin, Back & Body Chiropractic
Elspeth Cowell MSCh DpodM SRCh HCPC reg
William Hunter, Nuffield Health
Be at the leading edge of sports injury management
Our international team of qualified experts (see above) spend hours poring over scores of technical journals and medical papers that even the most interested professionals don't have time to read.
For 17 years, we've helped hard-working physiotherapists and sports professionals like you, overwhelmed by the vast amount of new research, bring science to their treatment. Sports Injury Bulletin is the ideal resource for practitioners too busy to cull through all the monthly journals to find meaningful and applicable studies.
*includes 3 coaching manuals
Get Inspired
All the latest techniques and approaches
Sports Injury Bulletin brings together a worldwide panel of experts – including physiotherapists, doctors, researchers and sports scientists. Together we deliver everything you need to help your clients avoid – or recover as quickly as possible from – injuries.
We strip away the scientific jargon and deliver you easy-to-follow training exercises, nutrition tips, psychological strategies and recovery programmes and exercises in plain English.










