Uncommon injuries: proximal hamstring rupture - act sooner rather than later
Chris Mallac looks at the etiology, diagnosis, and treatment options for proximal hamstring ruptures in athletes.
Southeast Asian Games 2019 Vietnam’s Nguyen Tung Duong in action during the Men’s 3m Springboard Final REUTERS/Athit Perawongmetha
Although an uncommon form of hamstring damage (only eight to 12% of all hamstring injuries), an untreated rupture at the muscle origin leads to significant functional debilitation(1-3). The actual incidence of undiagnosed rupture may be much higher, thus accounting for the many athletes who seem to have chronic hamstring problems(4). Athletes in their early to mid-forties who have a degree of underlying degeneration in the hamstring tendon are the most vulnerable(3,4). However, it does occur in younger athletes(5).
Anatomy and biomechanics
Four muscles comprise the hamstrings:
- Biceps femoris long head (BFLH)
- Biceps femoris short head (BFSH)
- Semitendinosus (ST)
- Semimembranosus (SM)
All four muscles participate in knee flexion, with the BFLH, SM, and ST also assist with hip extension (the BFSH is not active in hip extension). The ST and BFLH share a common origin on the anteromedial ischial tuberosity. The SM originates more proximally and posterolaterally on the ischial tuberosity (see figure 1)(6,7).
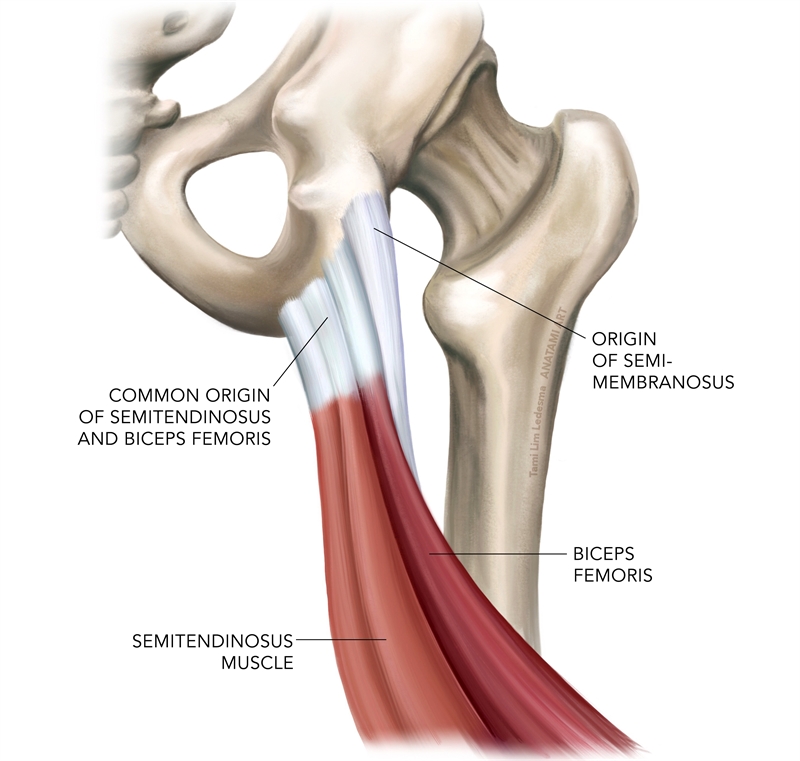
Figure 1: Hamstring origin anatomy
Epidemiology
The most likely mechanism for a proximal hamstring rupture is sudden knee extension with concurrent hip flexion while under an eccentric load. This motion happens in a slip or fall, and in sports such as water and snow skiing, bull riding, running, rugby and other football codes, dance, and gymnastics(2,8,9). The only significant intrinsic risk factor is pre-existing degeneration in the hamstring tendons, which likely accounts for the higher prevalence of hamstring rupture in over 40 recreational athletes(10). This injury in a young, skeletally immature athlete usually indicates an avulsion of the apophysis. The ischial apophysis does not fuse until the late teenage years or early twenties. During this time, the apophysis and bone are functionally weaker than the tendon(11,12).
Diagnosis
The signs and symptoms of a ruptured hamstring origin are not always clear and possibly mistaken for a muscle strain. However, some of the more definitive tell-tale signs and symptoms are(2,3,8,13,14):
- Sudden onset of sharp pain and the feeling of “being shot in the thigh.”
- Feeling or hearing a pop in the back of the leg.
- Antalgic gait with a stiff leg to avoid hip flexion and knee flexion.
- Marked bruising in the posterior thigh and gluteal region during the days following the injury.
- Pain on sitting on hard chairs and car seats.
- Pain when walking upstairs.
- Referred distal hamstring pain if it affects the sciatic nerve.
- Weakness in the involved hamstring.
- Pain and limited range of motion on straight leg raise.
- Palpable mass in the hamstring if significant tendon retraction.
- Foot drop may be present in the presence of peroneal nerve neuropraxia.
- Bowstring sign - an absence of palpable tension in the distal part of the hamstrings with the patient prone and the knee flexed to 90 degrees(13).
Imaging
Hamstring origin disruptions can be assessed adequately with skilled sonography. However, MRI is still the imaging modality of choice to ascertain the degree of retraction and the extent of tendon rupture (see figure 2)(1). MRI sequences should include T1, T2, inversion recovery, and fat-suppressed sagittal, axial, and coronal views. Coronal views ascertain the extent of retraction while the axial images identify which tendons are involved. Sagittal views show distal muscle injury(4). The clinical presentation and imaging assist in the classification of the severity of the rupture (see table 1).
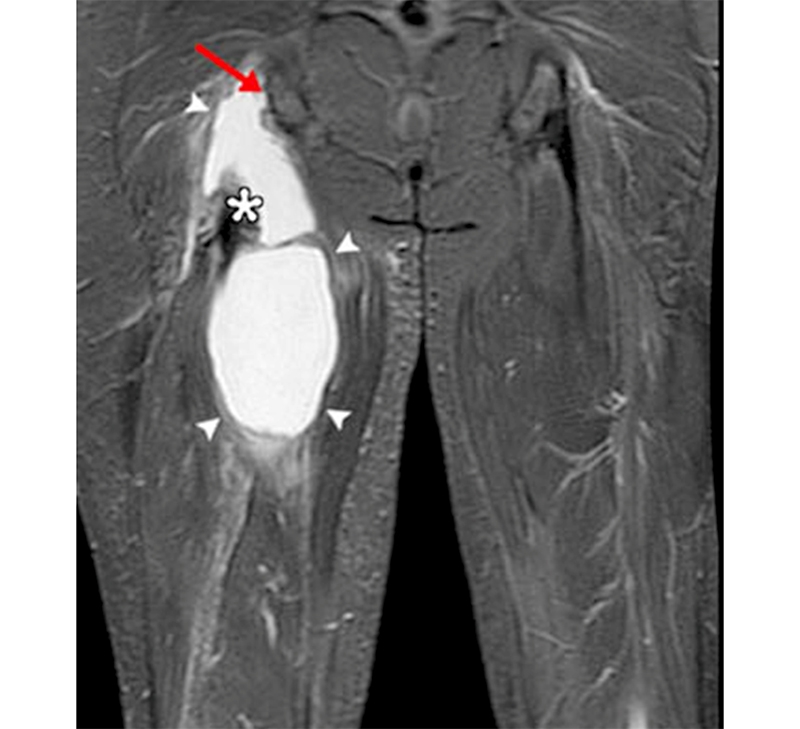
Figure 2: MRI showing complete rupture of the hamstring origin and associated large hematoma
Conservative management
In single-tendon tears, the ruptured tendon adheres to the remaining intact tendons to form a pseudo tendon of scar tissue(3). A single-tendon rupture that has only retracted one to two centimeters is well treated conservatively, and often athletes return to sport in six to eight weeks. Partial proximal hamstring tendon tears with less than two centimeters of retraction do well with either non-operative treatment or operative treatment(21). Therefore a trial of conservative management is appropriate for the first three months with partial tears.
Failed conservative management of hamstring avulsions results in pain with sitting and running, weakness in hip extension and knee flexion, poor endurance in the hamstring, and sciatic nerve complications(15). Consider a referral for surgical correction if these symptoms don’t significantly resolve after three months. Isokinetic testing shows that surgically repaired hamstrings return to 91% of the strength of the uninjured leg, whereas non-operated hamstrings only regained 57-67% of uninjured strength(22).
Table 1: The clinical classification and management of proximal hamstring injuries.
| Classification | Pathological appearance | Management |
|---|---|---|
| 1 | Osseous avulsions | Greater than 1-2cm retraction; need internal fixation |
| 2 | Musculotendinous junction injuries | Conservative treatment (as difficult to treat surgically) |
| 3 | Incomplete avulsions | Conservative treatment |
| 4 | Complete avulsion with minimal retraction | Depending on the degree of retraction (usually >2cm) may need surgery |
| 5a | Complete avulsion with retraction and no sciatic nerve involvement | Usually surgery |
| 5b | Complete avulsion with retraction with sciatic nerve involvement | Usually surgery |
Criteria for surgery
Research now recommends surgical management if patients meet the following criteria:
-
Two tendons rupture with more than two centimeters of retraction. (NB: surgical studies reveal that tendons may retract up to 10cm(15). Further observations show that when two tendons rupture and retract more than two centimeters, the musculotendinous junction of the remaining tendon may also suffer a significant injury(3).
-
Complete rupture of all three tendons with or without significant retraction.
-
Failed conservative treatment with persistent pain when sitting and an inability to run(16).
-
Unresolved loss of hamstring power and sciatic nerve involvement(15).
-
Avulsion of the ischial apophysis in the skeletally immature athlete(15).
Acute surgical repair (within four weeks of injury) of hamstring origin ruptures leads to improvements in functional outcomes, strength, and endurance in the muscle(2,3,5,9,17). Furthermore, acute surgery carries a low risk of complications and re-rupture and a high rate of return to the pre-injury level of sports(9).
Delayed surgical repairs are not as effective as acute repairs, yet they lead to more substantial improvements in strength, endurance, and return to pre-injury levels of sport than non-surgical management. However, while reasonably successful, delayed surgery is technically more difficult due to:
-
Sciatic nerve tethering (this may happen two to five weeks post-injury)(18).
-
Advancement of the retraction.
-
Possible fractional muscle lengthening.
-
Larger surgical incisions are needed(5,9).
-
Greater fatty deposits make reattachment more difficult due to the degradation of the tissue bed(14,15,19).
The impingement of the sciatic nerve via tethering to the hamstring origin, known as hamstring syndrome, produces pain in sitting or constant buttock pain. The pull of the retracted muscle on the motor branch of the sciatic nerve causes the pain. Some surgeons advocate automatic sciatic nerve exploration and neurolysis in all surgical cases to prevent post-operative nerve complications(5,15,20).
Post-surgical rehabilitation
With a delayed repair (six weeks or more post-injury), the patient is placed in a fixed knee-flexion brace to 90 degrees or a hip orthosis to limit hip flexion to 40 degrees(2,5). This limited range avoids stretching the shortened muscle and protects the reattachment at ischial tuberosity. The brace is gradually expanded to full knee extension over eight weeks as the hamstring accommodates to the stretch(8,15).
About 50% of patients experience some post-surgical neuralgia symptoms in the first 12 months post-surgery(2). Roughly half also complain of pain at the incision site when sitting(2). The typical post-operative rehabilitation phases are(17):
Acute healing phase (0-2 weeks)
-
- The focus is on pain and swelling control.
- No massage to the hamstring for the first four weeks.
- Core stability exercises commenced.
- Gentle neural mobility without placing strain on repair.
- Partial weight (25%) bearing on crutches with short steps.
- Avoid sitting on the repaired hamstring tendons.
- Isometric gluteal and quadriceps exercises.
- Ankle pumps to avoid DVT’s.
Continued healing and repair phase (2-6 weeks)
-
- Week two: begin passive range of motion of the knee and hip.
- Week four: active motion allowed at hip and knee.
- Active hip flexion in knee flexion (no resistance).
- Active knee extension in hip neutral (no resistance).
- Progress toward full weight-bearing with normal gait by week six.
Continued repair phase (6-12 weeks)
-
- Begin with isometric and progresses to concentric exercises – no resistance. Avoid heavy eccentric loading.
- Core strength and proprioception.
- Focus on gluteal strength.
- Avoid aggressive stretching for the first three months.
Remodeling phase (12-24 weeks)
-
- Start loaded hamstring exercises with focus on ‘feeling’ the hamstring via biofeedback (touch or EMG).
- Start a progressive eccentric-focus program.
- Regain full knee and hip motion.
- Resume running around week 16 (assuming 70% strength in repaired hamstring).
- Also, begin plyometric activities at week 16.
- Landing mechanics and deceleration exercises start at week 19.
Sports-specific phase (24+weeks)
-
- Start sprinting and change of direction activities with a slow return to skills.
The average time to return to sports after surgery is 6.6 to 8.5 months(2,3). The average return to competition timeline after early surgical repair is 16 weeks, 25 weeks in delayed surgeries, and 29 weeks in operations performed six months or more after injury. Full return to sports was nine weeks faster in early-repaired athletes compared to delayed repairs and 13 weeks faster than late repairs(5).
Summary
A ruptured hamstring origin is an infrequent but potentially catastrophic injury if left untreated. They are most often ruptured when the athlete suffers a rapid hip flexion with knee extension movement. Immediate surgical repair is required if one or two tendons rupture and retract more than two centimeters, all three tendons rupture, or avulsion occurs in a skeletally immature athlete. Athletes are candidates for delayed surgery if, after conservative treatment for three months, they still suffer weakness, reduced sprint speed, and pain with sitting. Rehabilitation protocols are reasonably standard, and athletes can return to sports anywhere from 16 to 29 weeks, depending on the timeliness of the surgery and the demands of their sport.
References
- Skeletal Radiol. 2003;32:582–589
- Am J Sports Med. 2012; 40:2092-2098
- Acad Orthop Surg. 2007;15:350-355
- Oper Tech Sports Med. 2009. 17:196-204
- The American Journal of Sports Medicine. 2015. 43(2): 385-391
- J Bone Joint Surg Am. 2007;89:44-48
- Magn Reson Imaging Clin N Am. 2005.13:677–690, vi, PMID:16275576
- Wood J Bone and Joint Surgery 2008. 90, 2365-2374
- Int J Sports Med. 2011; 32: 490 – 495
- Am J Sports Med. 2013;41(12):2933–47
- Emerg Med J. 1999;16: 457–8
- Ann R Coll Surg Engl. 2007; 89: 394–399
- J Bone and Joint Surgery. 2011. 93; 1819-1826
- Knee Surg Sports Trauma Arthroscopy. 2013. 21; 515-533
- The Journal of Bone and Joint Surgery 2009. 91-A, Supp 2, part 2. 249-256
- J Bone and Joint Surgery Br. 2012. 94; 660-662
- Aspetar Sports Medicine Journal.2019, 8, 60-65
- Int Orthop. 2010. 34:119–123
- Am J Sports Medicine. 2008. 36; 1110-1115
- Arthroscopy Techniques. 2017. 6(2), e311-e317
- The Orthopaedic Journal of Sports Medicine. 2017.5(2), 1-7
- The Orthopaedic Journal of Sports Medicine. 2017.5(11). 1-6
Newsletter Sign Up
Subscriber Testimonials
Dr. Alexandra Fandetti-Robin, Back & Body Chiropractic
Elspeth Cowell MSCh DpodM SRCh HCPC reg
William Hunter, Nuffield Health
Newsletter Sign Up
Coaches Testimonials
Dr. Alexandra Fandetti-Robin, Back & Body Chiropractic
Elspeth Cowell MSCh DpodM SRCh HCPC reg
William Hunter, Nuffield Health
Be at the leading edge of sports injury management
Our international team of qualified experts (see above) spend hours poring over scores of technical journals and medical papers that even the most interested professionals don't have time to read.
For 17 years, we've helped hard-working physiotherapists and sports professionals like you, overwhelmed by the vast amount of new research, bring science to their treatment. Sports Injury Bulletin is the ideal resource for practitioners too busy to cull through all the monthly journals to find meaningful and applicable studies.
*includes 3 coaching manuals
Get Inspired
All the latest techniques and approaches
Sports Injury Bulletin brings together a worldwide panel of experts – including physiotherapists, doctors, researchers and sports scientists. Together we deliver everything you need to help your clients avoid – or recover as quickly as possible from – injuries.
We strip away the scientific jargon and deliver you easy-to-follow training exercises, nutrition tips, psychological strategies and recovery programmes and exercises in plain English.