Blood Biomarkers for Screening Active Women
When clinicians assess physically active females, they must consider key issues, including low iron stores, vitamin D deficiency, and suppressed energy hormones, due to the real physiological risk. Cory Dugan and Hayley Carratti present a practical blood biomarker screening framework that coaches, physiotherapists, and active women can use today.
The Case for Screening
A semi-elite female athlete presents with persistent fatigue and a declining race time. Her general practitioner runs a standard blood panel and tells her everything looks normal. What is commonly missed: her serum ferritin (the blood marker that reflects iron stores) is 12 ng/mL, a level that sports medicine practitioners recognize as indicating iron deficiency requiring intervention, not only for performance but for overall health, wellbeing, and quality of life. Her coach attributes the fatigue to training load. Nothing changes. Her times continue to drop.
This scenario is not rare, and it highlights a persistent failure in the healthcare of active women. Clinicians often rely on general population reference ranges that do not account for active individuals, and coaches lack the tools to distinguish physiological deficiency from an inadequate training response. The consequence is a large population of women exercising for health, with no systematic framework for identifying the deficiencies that undermine it.
Screening is not optional in high-performance sport; it is foundational. Yet between the elite athlete who undergoes structured biomarker monitoring and the sedentary adult who receives a standard annual blood test, there exists a vast and largely unserved population: the semi-elite female athlete. She is not sedentary, so general population clinical thresholds may not reflect her physiology. She is not elite, so the monitoring infrastructure of professional sport is unavailable to her. She sits in a gap, and that gap has real consequences.
Monitoring Athlete Health
Clinicians can approach monitoring the health and physiological status of active women in several ways. Questionnaires and wellness scales capture subjective perceptions of fatigue, mood, and recovery readiness. Performance tests (timed runs, jump assessments, grip strength) provide objective but variable measures of neuromuscular status. Heart rate monitoring and wearable devices track acute internal load during training. Dietary assessment tools attempt to quantify energy and nutrient intake, though these are subject to significant reporting bias and require impractical levels of data collection to be reliable.
Each of these tools has value, and clinicians shouldn’t use them in isolation. However, all share a common limitation: they measure downstream consequences rather than upstream causes. By the time fatigue appears on a wellness scale or a performance test flags a decline, a physiological deficiency has often been present for weeks or months. Clinicians need a tool that can detect problems before they manifest as symptoms, and blood biomarker screening is a powerful option available for doing so.
"Clinicians need a tool that can detect problems before they manifest as symptoms..."
Blood Biomarker Screening
Proteins, metabolites, electrolytes, and other small molecules circulating in the blood serve as biomarkers (measurable indicators of physiological processes) for athletes and recreationally active individuals alike. Advances in data-driven approaches to health assessment suggest that biochemical and hematological (specifically blood-based) data can be powerful in identifying the balance between training stress and recovery in each unique individual. Many commercially available services now offer biomarker testing (although their validity and reliability are largely unknown), and professional athletes are increasingly exploiting this technology to track performance and recovery during training. Yet access to evidence-based, sex-specific, practically applicable guidance for non-elite active women remains limited.
Sex-specific Protocols
Female athletes have a fundamentally different physiological context from male athletes. Nutritional demands, hormonal fluctuations, and training stress interact in ways that standard, male-derived screening protocols fail to accurately track. Menstrual cycle hormonal fluctuations directly alter physiological responses to training loads(1). The same training stimulus elicits meaningfully different stress responses across distinct cycle phases, complicating performance adaptation and clinical monitoring.
The menstrual cycle introduces complexity that extends to the biomarkers themselves. Inflammation markers and stress hormones both shift across specific phases of the cycle. Two blood draws from the same woman at different points in her cycle will appear meaningfully different despite no true clinical change occurring. The oral contraceptive pill (OCP) introduces a further layer: by suppressing endogenous estradiol and progesterone production, it fundamentally changes the interpretation of the entire hormone panel, shifting diagnostic weight from reproductive hormones to bioenergetic markers such as thyroid hormone.
Active women of reproductive age also face a dual physiological burden: exercise-induced micronutrient losses compounded by regular menstrual blood loss. Iron deficiency prevalence in exercising women is estimated as high as 57% in some populations(2). Co-occurring deficiencies in vitamin D, vitamin B12, and folate are common, compounding fatigue through overlapping mechanisms. Low energy availability (LEA; a state where dietary intake is insufficient to meet both exercise demands and basic bodily functions) is an underappreciated additional risk that suppresses hormonal function, impairs bone health, and elevates injury risk(3).
The Blood Biomarker Screening Panel
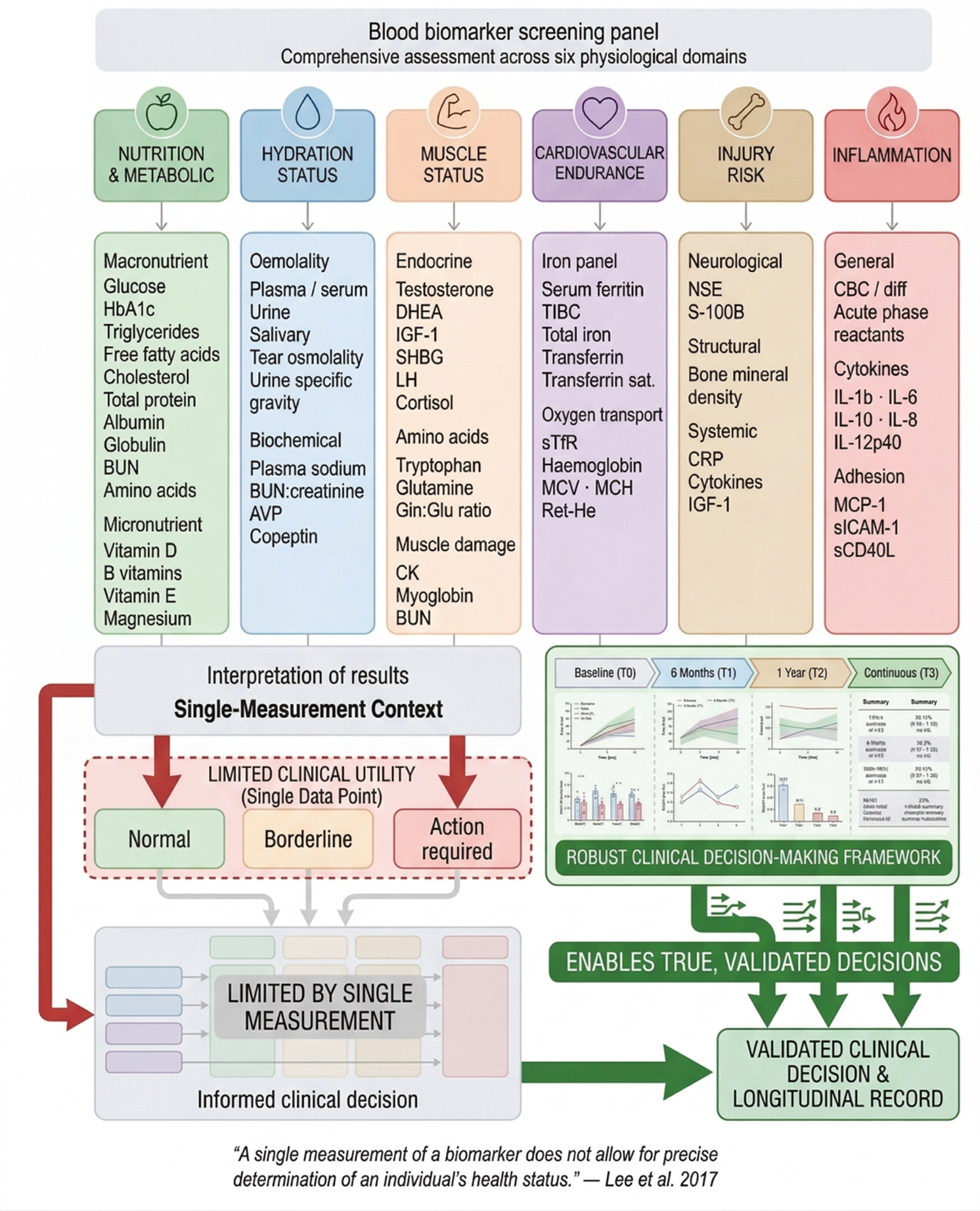
A blood biomarker screening framework may provide a solution for clinicians working with female athletes (see Figure 1)(3-6). The framework covers six physiological domains: nutrition and metabolic health, hydration status, muscle status, cardiovascular endurance, injury risk, and inflammation. Each domain contains the specific markers most relevant to assessment within it. The flow moves from the individual woman through the panel domains to an interpretation step, and finally to one of three action pathways, with a longitudinal record running in parallel.
Considerations for the Semi-elite Female Athlete
Not every marker in Figure 1 needs to be assessed in every screen, nor can every practitioner or athlete access the full panel. For the semi-elite female athlete, two practical realities shape every panel decision: financial cost and access. Comprehensive biomarker testing represents a meaningful investment, and a blanket reduction of the panel is not the right response to budget constraints. The smarter approach is to apply individualized risk stratification. Clinicians should determine the highest-yield markers for each individual athlete with some level of sophistication, as they will deliver the greatest clinical return and should be treated as non-negotiable regardless of budget. Then, clinicians can incorporate lower-priority additions when resources allow or when a specific clinical picture warrants them. Group testing through sports medicine clinics or pathology providers with athlete-specific service agreements can substantially reduce per-test cost. Access to venous blood collection also requires a qualified practitioner and a laboratory, and the blood draw volume and frequency should always be kept to a minimum, with a clearly justified purpose.
It is also worth distinguishing between two approaches to biomarker measurement: screening (infrequent testing, several months apart, to identify deficiencies or excesses) and monitoring (high-frequency testing, days or weeks apart, to track ongoing adaptation and recovery). For most semi-elite female athletes, screening is the realistic and appropriate starting point.
Underpinning all of these decisions is the principle of individualization. No two semi-elite female athletes present with the same combination of training history, menstrual health, dietary pattern, life stressors, and clinical history. A panel appropriate for one woman may overlook the most relevant deficiency in another. Clinicians need to evaluate the conditions that determine whether a screening program will succeed in practice and the suitability of any individual marker before it is included (see Table 1 and 2)(3). Frameworks are tools for building a defensible, individualized panel, not a universal checklist to be applied uniformly.
"Clinicians should determine the highest-yield markers for each individual athlete with some level of sophistication..."
Table 1: Key factors for the success of a biomarker screening program
|
Success factor |
Consideration for recreationally active women |
|
Clinical oversight and GP collaboration |
Essential: results flagging pathology require medical follow-up |
|
Appropriate actionable marker selection |
Choose markers that will change management, not just generate data |
|
Contextual data for interpretation |
Cycle phase, OCP use, recent illness, and training load must be documented |
|
Patient buy-in and clear feedback |
Results must be communicated in an accessible language to be acted upon |
|
Cost-benefit justification |
Tier selection should match clinical need and available budget |
|
Statistical approach to change detection |
Longitudinal within-individual comparison is more informative than population norms |
(adapted from Pedlar et al. 2019)
Table 2: Biomarker suitability checklist for selection decisions
|
Criterion |
Key question |
|
Evidence base |
Is this marker validated clinically, in sport, or in this specific population? |
|
Actionability |
Will this result change clinical management or advice? |
|
Validity |
Has it been validated in exercising women specifically? |
|
Variability |
Is the coefficient of variation acceptable for repeat testing? |
|
Collection practicality |
Can it be collected and processed in a clinic or GP setting? |
|
Diurnal variation |
Does time of day or cycle phase meaningfully affect the result? |
|
Cost |
Is the cost justified relative to the clinical yield for this population? |
|
Covariates |
Are OCP use, inflammation, and cycle phase documented to aid interpretation? |
(adapted from Pedlar et al. 2019)
Blood collection
A screening program is only as reliable as the conditions under which samples are collected. Poor pre-analytic control, the conditions surrounding the blood draw itself, is one of the most consistent sources of error in applied biomarker testing, yet it is frequently overlooked outside formal laboratory settings(4, 5). For the clinical setting, the practical pre-draw protocol checklist captures the essentials:
- Sample collected in the morning following an overnight fast,
- No high-intensity exercise in the 24 hours prior to collection,
- Seated posture for at least 10 minutes before venipuncture,
- Menstrual cycle phase documented: early follicular phase preferred for hormonal markers,
- OCP use recorded: suppresses sex steroids, shifting diagnostic weight to fT3 and IGF-1,
- Recent illness or injury noted: both elevate CRP and can artificially raise ferritin,
- Same sampling site used consistently if capillary collection (earlobe or fingertip).
Consistency matters more than perfection. The same pre-analytic conditions across screens are what make longitudinal comparison meaningful. When clinicians cannot achieve ideal conditions, they should document deviations so that anomalous results can be contextualized rather than misinterpreted.
Interpreting Results
Once results are returned, a fundamental interpretive challenge presents itself. A single biomarker result, read without context or longitudinal comparison, carries limited diagnostic value. Blood testing shares a limitation similar to other monitoring tools discussed earlier: it can confirm that something is already wrong, but only trends across time allow a problem to be caught before it becomes symptomatic and detrimental to performance. As researchers at the University of Connecticut observed, "a single measurement of a biomarker does not allow for precise determination of an individual’s health status”. A single result describes the present situation and nothing more; a series of results reveals the trajectory. Yet in practice, clinicians classify results against population-derived reference ranges into categories such as “normal, borderline, and action required". This approach is familiar and widely used, and for the female athlete, it is frequently inadequate. Population norms are derived from cross-sectional samples of the general population; they do not account for physiological variation introduced by the menstrual cycle, training status, or energy availability. And this is where the science currently fails the practitioner.
Research in this area has disproportionately focused on population-level thresholds (the arbitrary ‘lines in the sand’ that define ‘normal’ and ‘not-normal’) rather than on within-individual changes that are clinically meaningful. Clinicians shouldn’t interpret single markers in isolation. Researchers at St Mary’s University demonstrated that individualized reference ranges built from repeated testing substantially outperform population-based clinical thresholds for detecting meaningful change within an individual(4). Two data points collected months apart under consistent pre-analytic conditions are already more informative than one. Three or four, spanning a training year, begin to reveal the individual’s personal ranges, seasonal patterns, and responses to training load that no population norm can provide.
"A single biomarker result, read without context or longitudinal comparison, carries limited diagnostic value..."
A Contextual Example
Given the breadth of markers available and the considerations outlined above, the following example illustrates how clinicians may approach panel selection for a hypothetical composite athlete. She is 34 years old, runs four times per week, has followed a plant-based diet for 18 months, donates blood every three months, is a mother of two children under five, and reports heavy periods (see Figure 2). She has no current symptoms and is presenting for a baseline pre-season screen.
Four independent iron-loss pathways converge in this single individual, making her a useful teaching case, as the panel’s decisions follow directly from her profile.
For this athlete, the iron panel and complete blood count represent the highest clinical priority. Five independent iron-loss pathways converge in her profile: foot-strike hemolysis from running four times per week, absence of dietary heme iron from 18 months on a plant-based diet, repeated phlebotomy losses from donating blood every three months, postpartum iron depletion with two children under five, and heavy menstrual blood loss. An additional absorption barrier compounds this picture: post-exercise hepcidin elevation suppresses ferroportin-mediated iron uptake for several hours after the run, reducing the benefit of oral supplementation if timed incorrectly.
Essential tests to consider:
- The full iron panel (serum ferritin, hemoglobin, transferrin saturation, TIBC, and sTfR, where ferritin is borderline or inflammation is present) at baseline and at each subsequent screen while this risk profile remains.
- hs-CRP is included alongside ferritin, given that ferritin behaves as an acute-phase reactant and cannot be interpreted in isolation without a concurrent inflammation check.
- sTfR is particularly valuable here as it is not an acute phase reactant, providing interpretive independence from CRP when inflammation is suspected.
- Vitamin D and B12 warrant Tier 1 inclusion given their high deficiency prevalence in plant-based populations and straightforward treatment pathways.
- The sex hormone panel (LH, FSH, oestradiol, progesterone if not on OCP, DHEA, cortisol, and free T3 as an energy availability indicator).
Where finances permit, folate (co-assessed with B12 and iron), electrolytes, and kidney function markers (creatinine, eGFR, urea) form a strongly recommended Tier 2, the latter providing a baseline against which exercise-related changes in-season can be accurately interpreted rather than mistaken for pathology. Zinc and magnesium are the most clinically relevant Tier 3 additions given the plant-based diet, alongside fasting glucose and HbA1c to complete the metabolic picture.
Conclusion
Semi-elite female athletes occupy a gap that standard clinical screening was never designed to serve. The physiological demands of regular exercise, compounded by the biological realities of the reproductive lifespan, create a pattern of risk that a single blood test compared against a population norm will consistently fail to detect. The tools to do better already exist. The markers are validated, the evidence base is robust, and the required infrastructure consists of a GP referral and a well-structured panel informed by the practitioner. What has been missing is a framework that considers this population on its own terms: one that accounts for the menstrual cycle, treats population thresholds as a starting point rather than an answer, and makes longitudinal within-individual comparisons a standard practice. Coaches, physiotherapists, and sports medicine practitioners who work with active women are well placed to implement this framework.
References
- Int J Environ Res Public Health. 2021;18(4):1667. doi:10.3390/ijerph18041667
- J Womens Sports Med. 2024;4(2):9-18. doi:10.53646/gde3q059
- Sports Med Auckl Nz. 2019;49(Suppl 2):185-198. doi:10.1007/s40279-019-01158-x
- Strength Cond Res. 2017;31(10):2920-2937. doi:10.1519/JSC.0000000000002122
- Eur J Appl Physiol. 2019;119(7):1463-1478.
- Sports Med. 2023;53(7):1315-1333. doi:10.1007/s40279-023-01836-x
Newsletter Sign Up
Subscriber Testimonials
Dr. Alexandra Fandetti-Robin, Back & Body Chiropractic
Elspeth Cowell MSCh DpodM SRCh HCPC reg
William Hunter, Nuffield Health
Newsletter Sign Up
Coaches Testimonials
Dr. Alexandra Fandetti-Robin, Back & Body Chiropractic
Elspeth Cowell MSCh DpodM SRCh HCPC reg
William Hunter, Nuffield Health
Be at the leading edge of sports injury management
Our international team of qualified experts (see above) spend hours poring over scores of technical journals and medical papers that even the most interested professionals don't have time to read.
For 17 years, we've helped hard-working physiotherapists and sports professionals like you, overwhelmed by the vast amount of new research, bring science to their treatment. Sports Injury Bulletin is the ideal resource for practitioners too busy to cull through all the monthly journals to find meaningful and applicable studies.
*includes 3 coaching manuals
Get Inspired
All the latest techniques and approaches
Sports Injury Bulletin brings together a worldwide panel of experts – including physiotherapists, doctors, researchers and sports scientists. Together we deliver everything you need to help your clients avoid – or recover as quickly as possible from – injuries.
We strip away the scientific jargon and deliver you easy-to-follow training exercises, nutrition tips, psychological strategies and recovery programmes and exercises in plain English.