Tendonitis vs tendinosis: the inflammation vs degeneration debate
Alicia Filley looks at tendon injury – and the ways in which these conditions have been diagnosed and treated…
Team Jumbo–Visma’s Wout van Aert in action with riders during stage 2 Pool via REUTERS/Papon Bernard
Tendons are bands of tough connective tissue that connect muscles to bones. They conduct the force generated by the muscle to move the bone; therefore, they must be strong enough to endure the transmission of forces, yet flexible enough to, in some instances, act as pulleys around bony prominences. Collagen, a protein found in the extracellular matrix (ECM) of connective tissue, gives tendons their tensile strength and allows them to be stretched without breaking. Collagen molecules bind together to form a microfibril, and microfibrils join one another to form a collagen fiber (see figure 1). Many collagen fibers make up a fiber bundle, and many fiber bundles joined together are called a tendon fascicle.
The fiber bundles and tendon fascicles are sheathed in a thin layer of loose connective tissue called the endotenon. The endotenon enables the bundles and fascicles to move independently of one another, sliding against each other as needed to adjust to the angle and force between the muscle and the bone. The endotenon is a continuation of the connective tissue that surrounds the entire tendon, called the epitenon. Some tendons have an additional sheath-like covering called the peratenon, which, while functioning with the tendon, is a distinct structure.
The tenocyte, the regulatory cell within the tendon, modulates the secretion of the extracellular matrix and the assembly of the collagen within the tendon. Tenocytes lie in long rows along the collagen fibers and are also found in the endotenon and epitenon of the tendon. The tenocytes form a connective web of finger-like projections that allows the cells to communicate with each other as to the need for synthesis or degradation of collagen fibres. They trigger the formation of more collagen cells when they experience stresses of short duration. Prolonged tension, however, results in collagen inhibition.
The blood supply to tendons is markedly less than that of the muscle to which they are attached. The vessels that do exist within the tendon are quite small, and run alongside the fascicles within the endotenon sheath. Some areas of tendon lack a blood supply altogether. As one might suspect, these areas are especially vulnerable to degeneration and rupture.
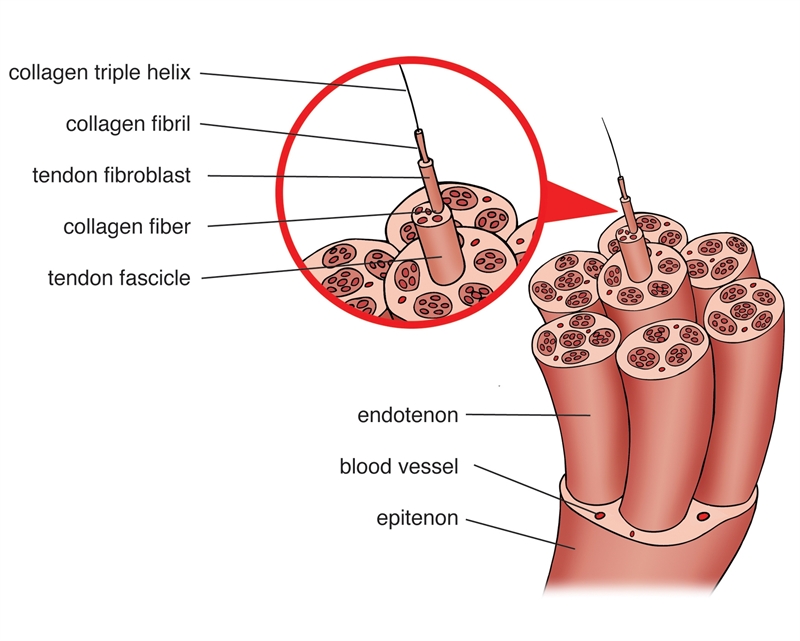
Figure 1: The anatomic structure of a tendon
Tendons are comprised of groupings of building blocks beginning with collagen molecules which join to form collagen microfibrils, then tendon fibroblasts, then fibers, fiber bundles, and fascicles. Fascicles are surrounded by the endotenon and grouped together to form the tendon itself.Tendon injury
Athletes frequently injure their tendons due to either overuse or trauma. Of the 32 million musculoskeletal injuries documented in the United States annually, 45% of them are injuries to tendons, ligaments, or joint capsules(1). The tendons most commonly injured by athletes are the tendons of the rotator cuff of the shoulder, the Achilles tendon, patellar tendon, and elbow extensor tendon.
Factors that place a strain on the tendon and contribute to overuse injuries include:
- Abnormal direction of pull due to skeletal misalignment;
- Differences in limb lengths;
- Muscle weakness or imbalances;
- Hypermobile joints;
- Inflexible muscles;
- Training errors;
- Faulty or improperly fitted equipment and shoes.
Notoriously difficult to treat, tendon injury was historically thought of as an inflammatory condition, and thus termed tendonitis. Treatment was aimed at reducing the inflammation through traditional anti-inflammatory medications and modalities, and was largely unsuccessful. Further study revealed that acute inflammatory cells were absent despite a disruption to the collagen formation within an injured tendon. A new term, tendinosis, was heralded to describe the degenerative lesions observed in the tendon tissue, and the absence of inflammation. Despite the new nomenclature and the addition of therapies to address the degeneration of the tendon rather than the inflammation, successful treatment of tendon dysfunction, which we shall call tendinopathy, has been elusive.
Inflammation plays a role
Advances in histology allow scientists to now take a closer look at the process of tendinopathy. Studies of injured human tendon are difficult because by the time a person seeks medical help the injury is usually chronic. Therefore, animal models are studied to reveal acute tendon changes. Scientists from Queen Mary University in London examined the response of the tenocytes of horse tendons to cyclic loading. Fascicle bundles from six horses were divided into treatment and control groups. Treatment samples underwent repeated loading strain, while controls remained unloaded.
Twenty-four hours after a cyclic loading protocol, the collagen cells within the fascicles of the treated tendons appeared rounded and unorganized, while the control cells were long, thin, and aligned longitudinally along the fascicle. Inflammatory markers were found in the treated samples after their loading cycle, while the control samples exhibited few if any markers for inflammation. Researchers concluded that tendon cells respond to high levels of stress with an inflammatory reaction, especially in the acute period following injury.
These findings are consistent with other animal studies, which find both an increase in inflammatory markers after episodes of loading or tendon damage, as well as an increase in the number and size of the tenocytes(2). The proliferation of tenocytes is known to occur in the presence of inflammation; therefore, this reaction is seen as an artifact of a previous inflammatory cascade(3). While degeneration is seen in chronic tendon injury, inflammation may be the instigator of those changes within the tendon during the acute period of tendon disease.
Further evidence
Ultrasound examination of symptomatic tendons shows an increase in blood flow to the tendons. Healthy tendons are characteristically lacking in blood supply. Therefore, to achieve this increased circulation, new blood vessels infiltrate the tendon. This neovascularisation typically occurs in conjunction with an accompanying nerve alongside the blood vessel. The sprouting of new nerves within the injured tendon is thought to be the source of pain in tendinopathy.
The influx of blood flow is assumed to be evidence of degeneration within the tendon and an attempt at healing the damaged tissue. However, such neovascularization and neoinnervation could likely not occur without the presence of inflammatory mediators at some point(4). Researchers at Cambridge University point to the fact that the appearance of the tendon body of patients with tendinopathy due to overload or injury is indistinguishable on ultrasound from those of patients with known inflammatory diagnoses such as rheumatoid arthritis.
Biochemical influences
Cyclooxygenase-2 (COX-2) is an enzyme, which in the presence of arachidonic acid, stimulates the production of prostanoids and produces inflammation. Studies show that levels of prostanoids are increased in animal tendons subjected to repeated loading(4). In tendons subjected to injected prostanoids, changes observed within the tendon are consistent with tendinopathy(3). Therefore, the presence of greater levels of prostanoids in diseased tendons is evidence of an inflammatory process within the tendon.
Substance P is a peptide secreted by nerves and inflammatory cells. The presence of substance P in significant amounts in chronic tendinopathy is thought to be the result of an inflammatory process within the tendon. Substance P causes an increase in the number of tenocytes in a tendon. Therefore, the increased number of tenocytes observed in an injured tendon may be the result of inflammatory mediators such as substance P. Substance P also increases the ratio of collagen III to collagen I molecules in the extracellular matrix (ECM). In a healthy tendon, collagen I is the predominant type found within the ECM. This change in the composition of the collagen in the ECM may well account for the difference in the shape and size of the collagen and the simultaneous disorganization observed in the London study(3).
Degeneration theory
Researchers in Melbourne, Australia, developed a multi-stage model of tendon injury that encompasses the current thinking on tendinosis (see figure 2)(4). When a healthy tendon experiences an increased load, it responds by increasing its stiffness to handle the greater force demand and increasing the production of collagen cells. The Australian researchers propose that this short-term proliferative but non-inflammatory cell response, is a reactive tendinopathy. Their thought is that the increase in cells is an attempt by the tendon to increase the cross-sectional area and, therefore, better handle the load. This short-term adaptation is reversible if the load is diminished or the tendon has a chance to rest before the next stress is applied. A healthy tendon adapts to the stress by growing larger and thus stronger. A diseased tendon does not recover from the stress and progresses to stage two.
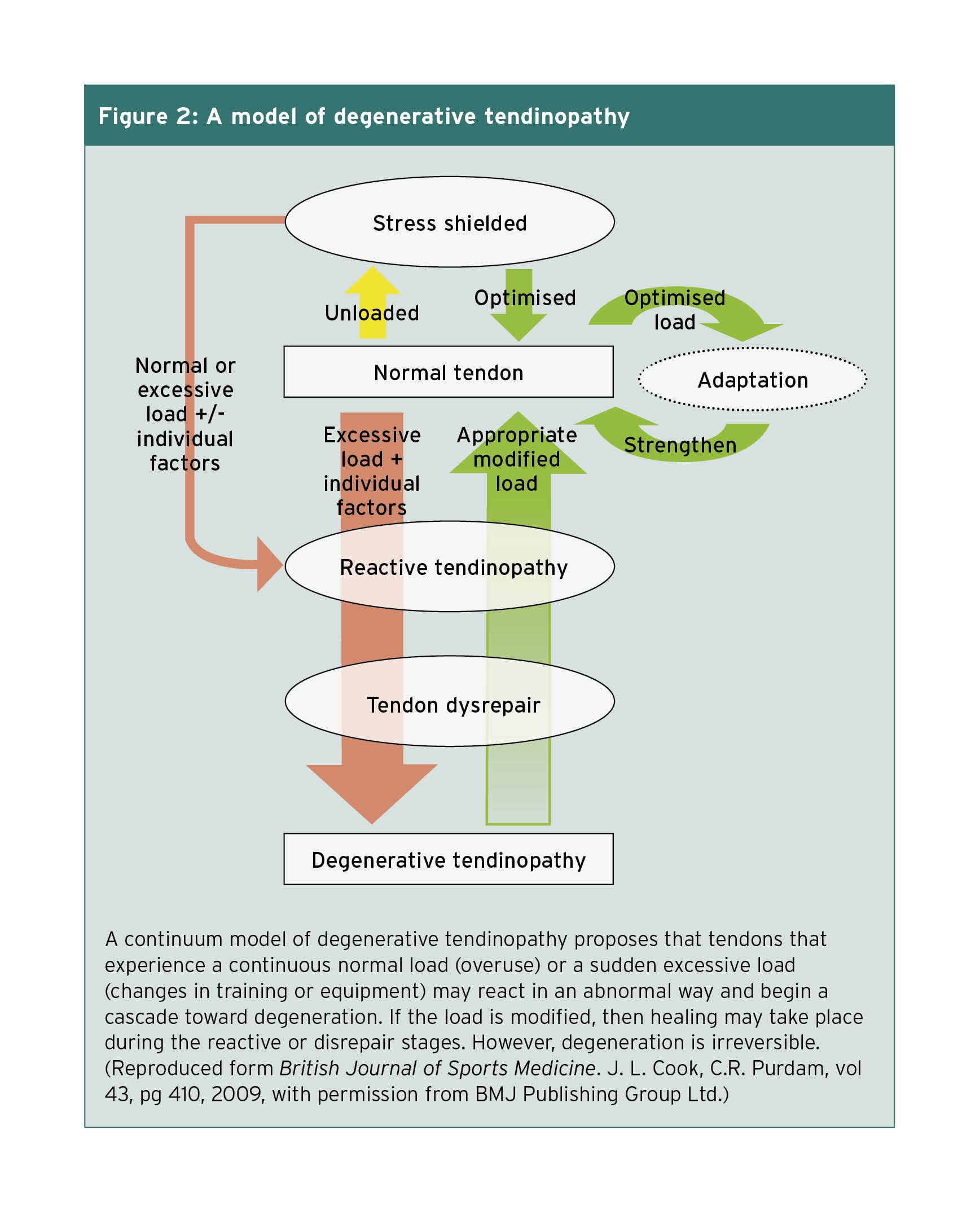
Figure 2: A model of degenerative tendinopathy
In the second stage, termed tendon disrepair, the tendon attempts to heal itself by adding more cells to the ECM, which increases the protein production of proteoglycans and collagen. They propose that it is the increase in the number of proteoglycans that changes the collagen composition and appearance and gives the ECM a more disorganized appearance. They note the increased vascularity at this stage. Their position is that with modification of the load applied to the tendon, the composition of the ECM can be altered and healing may still take place at this stage.
The final stage is degenerative tendinopathy. The hallmark of this stage is cell death, with areas of tendon completely devoid of healthy cells and an ECM filled with vessels, metabolic by-products, and little else. This stage is considered irreversible. Degenerative tendinopathy is found as distinct lesions within a tendon. The injured tendon may exhibit varying stages of degeneration throughout the tendon.
Two sides of the same coin
What histology reveals about injured tendons, in fact, is that inflammatory changes and degenerative changes are both found within the same tendon(5). A group of scientists from Italy and Sweden proposed a different model for tendinopathy, encompassing both inflammatory and degenerative observations. Termed the “iceberg theory”, this model begins with the assumption that normal exercise stimulates the production of new collagen within a tendon (see figure 3)(5). At the same time, collagen degradation also occurs, likely as a means of remodeling the tendon to accommodate the new load(6). Exercise, therefore, stimulates the production of inflammatory markers and growth substances, both needed for stimulation of the healthy tendon response of proliferation and degradation. In healthy tendons, collagen synthesis wins out in this equation and the tendon becomes larger and stronger.
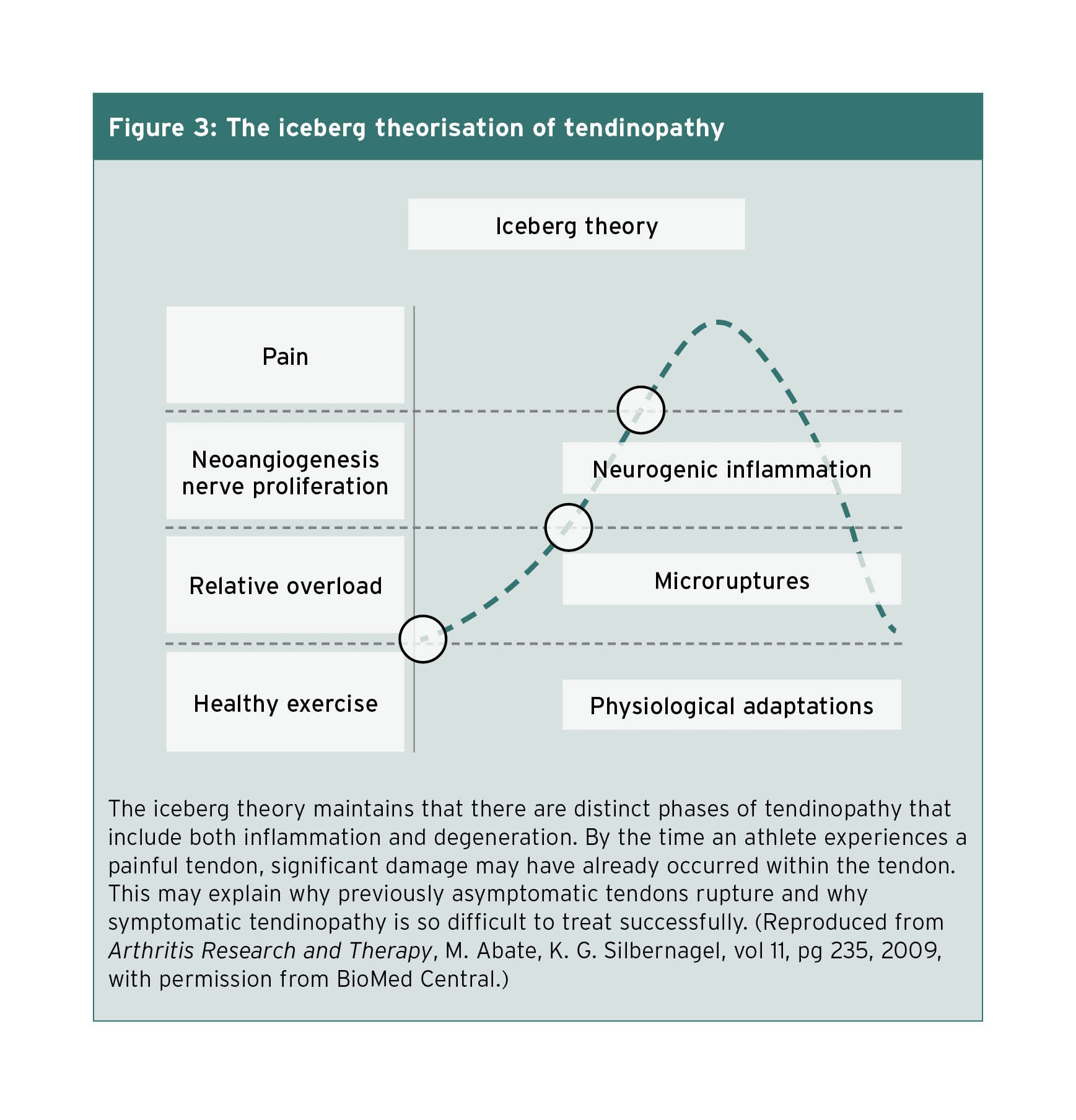
Figure 3: The iceberg theorization of tendinopathy
Cessation of loading, rest, and adequate blood supply are needed for the tendon to heal from excessive strain. If the tendon does not have the required blood supply, factors are produced which stimulate angiogenesis. The appearance of new vessels, which typically include nerves alongside the blood vessels, is thought to weaken the structure of the tendon(5). The secretion of both glutamate and substance P by the sprouting nerves contributes to a neurogenic inflammation as well as tendon cell death. It is at this point in the continuum that athletes may complain of pain and seek medical attention.
Clinical relevance
Understanding that there may be both inflammatory and degenerative processes in chronic tendinopathy may improve treatment outcomes. Since inflammation occurs early in the course of tendinopathy, non-steroidal anti-inflammatory drugs (NSAIDS) and steroid injections may be most effective at the onset of pain or when the athlete first notices a ‘tweaking’ or strain of the tendon. Sclerosing therapy and eccentric exercise both function to destroy or reduce the number of new blood vessels and nerves in the tendon. In theory, by reducing the number of new vessels, the tendon returns to normal and pain decreases. Eccentric exercise has the added benefit of stimulating collagen production(4). Manual therapies, such as augmented soft tissue mobilization, are thought to also stimulate collagen production and return the ratio of type III collagen to type I collagen within the ECM to normal.
This new paradigm of tendon disease stimulates new ideas for treatments. Treatments currently under investigation include biologic therapy, nitrous oxide, biochemical scaffolding, exogenous growth factor, platelet-rich plasma injection, stem cell injection, and tissue engineering. More research is needed to isolate which tendons, in what stage of disease, respond best to which therapy. In the meantime, the best recommendation is to treat any twinge or suspicion of tendinopathy early when the anti-inflammatory methods are most effective and the chance for healing is greatest.
You need to be logged in to continue reading.
Please register for limited access or take a 30-day risk-free trial of Sports Injury Bulletin to experience the full benefits of a subscription. TAKE A RISK-FREE TRIAL
TAKE A RISK-FREE TRIAL
Newsletter Sign Up
Subscriber Testimonials
Dr. Alexandra Fandetti-Robin, Back & Body Chiropractic
Elspeth Cowell MSCh DpodM SRCh HCPC reg
William Hunter, Nuffield Health
Newsletter Sign Up
Coaches Testimonials
Dr. Alexandra Fandetti-Robin, Back & Body Chiropractic
Elspeth Cowell MSCh DpodM SRCh HCPC reg
William Hunter, Nuffield Health
Be at the leading edge of sports injury management
Our international team of qualified experts (see above) spend hours poring over scores of technical journals and medical papers that even the most interested professionals don't have time to read.
For 17 years, we've helped hard-working physiotherapists and sports professionals like you, overwhelmed by the vast amount of new research, bring science to their treatment. Sports Injury Bulletin is the ideal resource for practitioners too busy to cull through all the monthly journals to find meaningful and applicable studies.
*includes 3 coaching manuals
Get Inspired
All the latest techniques and approaches
Sports Injury Bulletin brings together a worldwide panel of experts – including physiotherapists, doctors, researchers and sports scientists. Together we deliver everything you need to help your clients avoid – or recover as quickly as possible from – injuries.
We strip away the scientific jargon and deliver you easy-to-follow training exercises, nutrition tips, psychological strategies and recovery programmes and exercises in plain English.










