You are viewing 1 of your 1 free articles
The Controversial Anterolateral Knee Complex - Part 1
In the first of a two-part series, Alicia Filley unravels the mystery of the role of the ACL and anterolateral complex in knee stability. In the follow up article, she will build on these findings and evaluate the current thinking on conservative management, repair, and the required rehabilitation to successfully return athletes to sport.
The most common ligamentous injury in the knee - an anterior cruciate ligament (ACL) tear - is getting a sideways glance from researchers. What set off this scrutiny is the ‘failure’ rate of ACL reconstruction, with 25% of post procedure knees demonstrating rotational instability, up to 25% of young athletes suffering a second ACL injury, and just 55% of athletes returning to a competitive level of play (1,2).This frustration has led researchers to re-examine the anterolateral knee complex and the role of other structures in lateral and rotational stability.
What’s old is new again
In 2013, researchers in Belgium examined the anterolateral knee complex in 41 embalmed cadaveric knees(3). They identified the presence of a ‘new’ ligament they called the alterolateral ligament (ALL) in all but one specimen. They hypothesised that this ligament helped control internal tibial rotation and contributed to knee stability. Perhaps this was the missing link that explained why knees post ACL repair still demonstrated pivot shift laxity? (see Figures 1a-c) This set off a firestorm of research to both validate and refute the very existence of the ALL and its role in knee stability.
The existence of the ALL is actually not a ‘new’ phenomenon, but rather a rediscovery. French surgeon Paul Segond first identified the ALL in 1879 when he described a fracture of a small piece of bone from the lateral rim of the proximal tibia. He noted that the bone was avulsed by a shiny fibrous band on the anterolateral capsule of the knee. From that time, this lateral thickening was called by several different names in the medical literature, such as the anterior band of the lateral collateral ligament, lateral capsular ligament, or anterior oblique band.
In the 1970s, researchers correlated a ‘Segond fracture’ (easily visible on X-ray), with an ACL injury. Despite this resurgence of recognition, the role of the ALL in ACL injury and knee stability was overshadowed by the advent of knee arthroscopy in the later half of that decade. Even with the high success rate of technically correct arthroscopic procedures to repair the ACL (75% to 97%), knee instability remained a problem.
Tests for ACL integrity
*Note that clinicians should perform tests on both sides and record differences between the movement of the injured side versus the uninjured side.
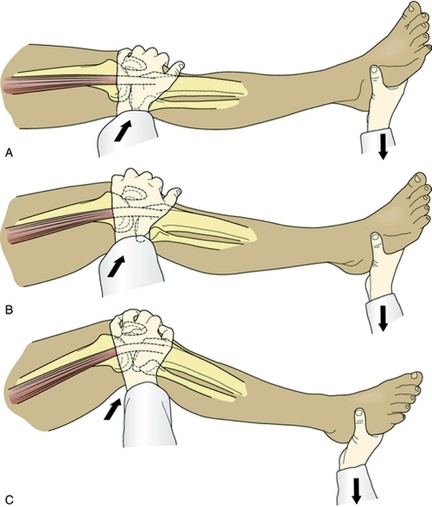
Pivot shift test(4) (Figure 1a below)
- Note that the patient’s knee is fully extended. Internally rotate the leg and apply a valgus stress.
- As the clinician flexes the knee between 20 and 45 degrees, the lateral tibial plateau is subluxed.
- As tension in the iliotibial band is lessened at 45 degrees of flexion, a pivot shift is felt as the tibia is reduced. This test identifies a rupture of the anterior cruciate ligament. Note that some knees demonstrate a positive pivot shift, even with a surgically corrected ACL(4).
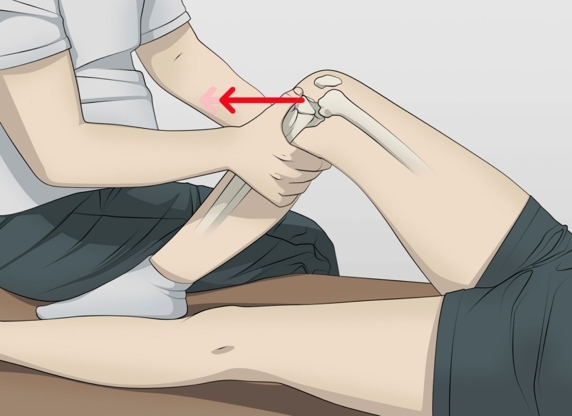
Anterior drawer test (Figure 1b above)
Position the patient in supine. Bend the knee to 90 degrees and stabilise the foot by partially sitting on it. Grasp the tibia with the both hands and pull forward. Excessive translation of the tibia or lack of an end feel indicates a positive test for a compromised ACL.
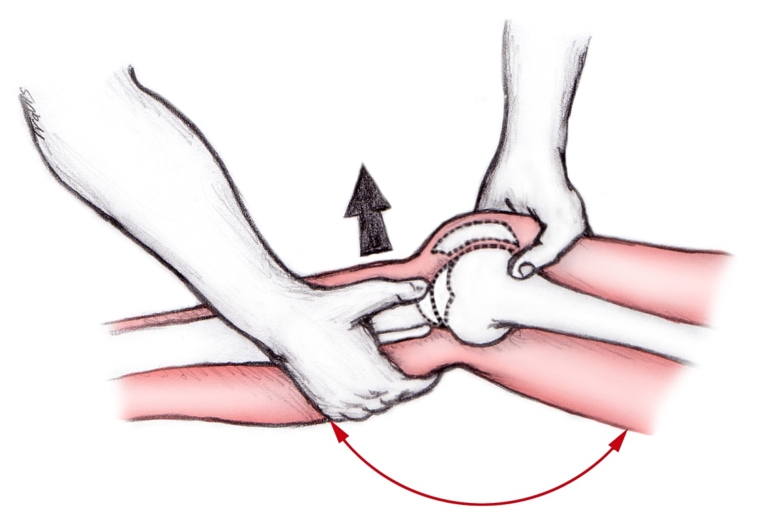
Lachman test (Figure 1c below)
Similar to the anterior drawer test, the Lachman test is conducted in supine with the knee flexed to only 25-30 degrees (red angle). The clinician stabilises the femur with the outside hand and grasps the tibia with the inside hand. Pulling the tibia anteriorly, an excessive excursion or lack of end feel is a positive result for ACL compromise.
Tunnel vision
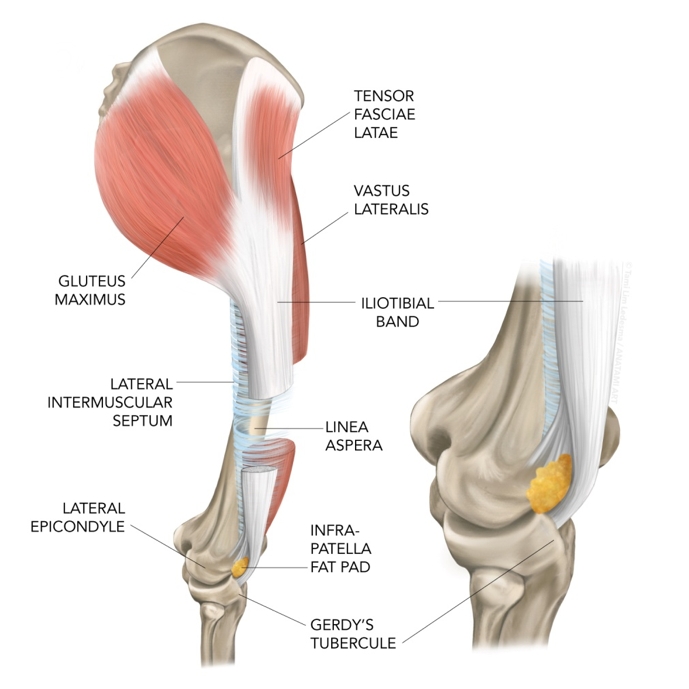
The knee joint consists of the femur and the tibia held together by ligaments. The lateral knee has three layers of stability: superficial, extracapsular, and intracapsular. Drilling down to the joint from the lateral side, one observes the superficial, medial and deep layers of the illiotibial band (ITB). This fibrous band of tissue arises from the tensor fascia lata at the pelvis and courses superficially along the lateral side of the thigh (see Figure 2).
The most superficial structure on the anterolateral knee is the illiotibial band (ITB). It arises from the pelvis, attaches to the posterior femur at the linea aspera via the intermuscular septum, and anchors with deep fibres both above and below the lateral joint line at the femoral epicondyle and Gerdy’s tubercle. Some propose a deeper layer called the capsulo-osseous layer with similar insertion sites.
Despite being the most lateral muscle, the ITB’s deep layer attaches, via the intermuscular septum, along the posterior femur at the linea aspera. The ITB anchors to the femur at the lateral epicondyle with a fan of fibres called Kaplan fibres, and to the tibia at Gerdy’s tubercle. Some maintain that the ITB has a deeper layer still, called the capsulo-osseous layer. It is proposed to lie just outside the joint capsule, arise at the superior border of the lateral femoral epicondyle, and insert posterior to Gerdy’s tubercle.
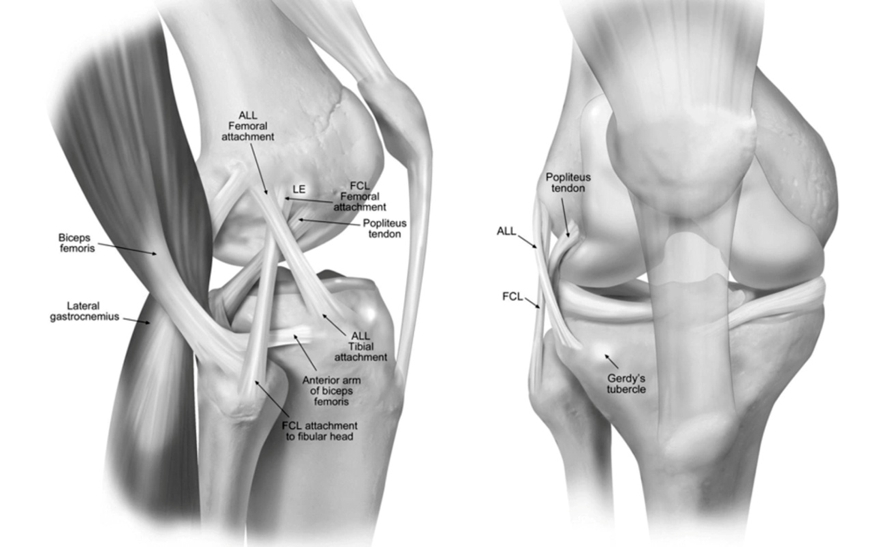
Outside the joint capsule, but deep to the ITB, the femoral collateral ligament (FCL) (sometimes called the lateral collateral ligament) spans from the lateral femoral epicondyle to the head of the fibula, where it joins the biceps femoris tendon. The ALL, identified outside the joint capsule on cadaveric specimens by the Belgium researchers, originates on the lateral femoral epicondyle and courses obliquely toward the anterolateral tibia, where it inserts midway between Gerdy’s tubercle and the fibular head. Within the joint capsule itself, the ACL arises from the posteromedial portion of the lateral femoral condyle and courses inferiorly, medially and anteriorly to the anteromedial aspect of the tibial plateau (see Figure 3)(3).
As described by Claes, et al (3). ALL, anterolateral ligament; LE, lateral femoral epicondyle; GT, Gerdy’s tubercle; FCL, femoral or lateral collateral ligament. Note the ALL arises from the LFE and inserts between Gerdy’s tubercle and the fibular head.
As interest grew in the structures of the anterolateral knee after 2013, many researchers went looking for the ALL themselves. While some confirmed the presence and location of the ALL, others felt it was not a ligament at all, but rather a thickening of the joint capsule. They termed this the ‘mid-third capsular ligament’. Still others were unable to locate any thickening, but noted deep fibres from the ITB, (the capsulo-osseous layer) were integrated with both the joint capsule and the bone of the tibia (4). How can there be so much scrutiny and yet so much confusion?
The main reason researchers disagree on the existence of the ALL has to do with the dissection technique they used. The appearance of cadaveric specimens differs depending on the preparation (frozen or embalmed). Some specimens were explored with the knee extended, while others were flexed and forcibly internally rotated, pulling the tissue on the anterolateral side of the knee taught. Therefore, researchers came away with conflicting views on the whether there existed a separate ligamentous structure (the ALL), a thickening of the joint capsule they call the mid-third capsular ligament, or a capsulo-osseous layer of the deep illiotibial band (ITB).
Moving target
The motion of the knee joint is complex, both gliding and rolling around an ever changing axis as the knee bends and straightens. The central location of the ACL within the joint allows it to function primarily as a tether between the tibia and the femur, preventing the tibia from moving forward out from under the femur. Due to the pull of the ACL, the tibia externally rotates in knee extension and internally rotates in flexion. Known as the ‘screw home’ mechanism, this tightening and pulling of the tibia into external rotation between 0°and 20°of flexion locks the knee, giving it greater stability in standing. When flexed beyond 20°, the ACL slackens and the tibia internally rotates. Therefore, some have questioned how the ACL alone controls tibial rotation and anterior excursion, especially in flexion?
Using a robot to test the resistance of cadaveric specimens to internal tibial rotation, a multicentre study measured the forces the ACL and anterolateral structures were able to withstand (4). At 30 degrees of knee flexion, the ITB resisted 44% of the internal rotation torque on the tibia; the ALL resisted 11%; the anterolateral capsule 3%; the ACL 9%; the medial collateral ligament 15%; and the posteromedial capsule 15%. This study therefore showed the ITB to be the main stabiliser in a knee flexed to 30 degrees.
This research seems to contradict previous studies, which showed the ALL to be instrumental in controlling internal tibial rotation(1). However, those studies were conducted with the knee at greater flexion angles or in ACL deficient knee. Given that as the knee flexes, the ACL slackens and ‘unlocks’ the tibia, it makes sense that the anterolateral structures would be recruited to modulate the resulting internal tibial rotation. Cadaveric tests show that the ALL tolerates loads of 50 to 320N - much less than the ITB which bears up to 769N. This again seems to point to the ITB as being better able to resist tibial rotation(4).
Missing the forest for the trees
The original question that sent many a researcher down the rabbit hole was why do so many technically correct and well executed ACL repairs still demonstrate rotational instability? Some look to a deficient ALL as the culprit. Box 1 summarises what is currently known about the ALL.
| Box 1: Summary of what is known about the ALL |
| Researchers disagree on its very existence. |
| It is a very small structure. |
| It withstands relatively small forces. |
| The presence of a Segond fracture correlates strongly with an ACL injury, therefore, it is assumed that the ALL plays a role in resisting internal tibial rotation. |
| Any resistance to internal tibial rotation it provides happens at greater than 30 degrees of knee flexion. |
The basic biomechanical principle that should be called to mind here is “big structures, big function, small structures, small function.”(4) Recall that the nearest ‘big’ structure, the ITB, originates at the pelvis, and courses superficially to the knee joint. Its deep fibres merge with the intramuscular septum, and attach to the linea aspera along the posterior side of the femur and anchor - via the Kaplan fibres - to the lateral femoral epicondyle, the joint capsule, and Gerdy’s tubercle.
The ITB, therefore, generates a line of pull posteriorly and laterally on the tibia as it forms a ‘sling’ around the posterior lateral epicondyle(4). The orientation of the ACL joins with these deep fibres of the ITB to nestle the lateral femoral condyle into an inverted horseshoe - thus keeping the tibia from moving forward beyond the femur. Doesn’t it therefore make more sense that the ITB - with deep capsule-osseous fibres in proximity to where the ALL is identified, a larger mass, an ability to withstand large forces, and a posterolateral line of pull - would be more likely to avulse a small piece of bone?
ACL testing anomalies
Surgeons in Columbus, Georgia evaluated 82 patients with acute knee instability using standard clinical tests performed under anaesthesia(5). They examined both intracapsular and extracapsular structures in surgery and correlated the severity of the observed injuries with the grade of the clinical tests. Ninety-eight percent of the knees (80 knees) presented with ACL tears. However, the significance of the tear did not correlate with the grading of the clinical tests. In other words, one would expect a more severe ACL tear to result in a more pronounced abnormality in testing, but this was not the case.
They also noted injury to the deep fibres of the ITB in 93% of the knees (76 knees) examined. These injuries DID correlate strongly with grades of abnormality in the clinical tests. Therefore, the examiners pointed out that traditional clinical examinations - when interpreted as only detecting deficiency in the ACL - miss the complete picture. They further asserted that variations in grades of the pivot shift and Lachman test are particularly sensitive at detecting the extent of injury to the deep fibres of the ITB, such that a more abnormal test signifies a more extensive injury.
Surgeons, who examined live specimens a full 20 years before the Belgium researchers resurrected the question of the ALL, concluded that the capsulo-osseous fibres of the ITB function as an anterolateral ligament. Therefore, it’s possible that the whole conundrum of the anterolateral knee anatomy comes down to variations in dissection technique and semantics. When it comes to function, however, it makes sense that deep insertions of the ITB work synergistically with the ACL to control anterior translation and internal tibial rotation.
Whether you call these fibres the ALL, the capsulo-osseous portion of the ITB, or the mid-third capsular ligament, it’s clear that the ACL does not control internal tibial rotation and anterior tibial movement alone. This synergy between the structures of the anterolateral knee and the ACL is such that the degree of injury to the ITB, not the ACL, correlates strongly with the grade of instability in clinical tests. Therefore, when these tests continue to yield abnormal results even after ACL reconstruction, the deep fibres of the ITB may still be impaired. What this means for intervention and rehabilitation will be explored in part II of this series.
References
- Clin Sports Med. 2018 Jan;37(1):33-40
- EFORT Open Rev. 2017;2:410-20
- J Anat. 2013;223:321-8
- Clin Sports Med. 2018 Jan;37(1):21-31.
- Am J Sports Med. 1993 Jan-Feb;21(1):55-60
Newsletter Sign Up
Subscriber Testimonials
Dr. Alexandra Fandetti-Robin, Back & Body Chiropractic
Elspeth Cowell MSCh DpodM SRCh HCPC reg
William Hunter, Nuffield Health
Newsletter Sign Up
Coaches Testimonials
Dr. Alexandra Fandetti-Robin, Back & Body Chiropractic
Elspeth Cowell MSCh DpodM SRCh HCPC reg
William Hunter, Nuffield Health
Be at the leading edge of sports injury management
Our international team of qualified experts (see above) spend hours poring over scores of technical journals and medical papers that even the most interested professionals don't have time to read.
For 17 years, we've helped hard-working physiotherapists and sports professionals like you, overwhelmed by the vast amount of new research, bring science to their treatment. Sports Injury Bulletin is the ideal resource for practitioners too busy to cull through all the monthly journals to find meaningful and applicable studies.
*includes 3 coaching manuals
Get Inspired
All the latest techniques and approaches
Sports Injury Bulletin brings together a worldwide panel of experts – including physiotherapists, doctors, researchers and sports scientists. Together we deliver everything you need to help your clients avoid – or recover as quickly as possible from – injuries.
We strip away the scientific jargon and deliver you easy-to-follow training exercises, nutrition tips, psychological strategies and recovery programmes and exercises in plain English.