Management of high hamstring tendinopathies — a conservative versus injection therapy approach
Trevor Langford reviews the possible ways to treat these hamstring problems – and the evidence on offer.

Jermaine Pennant holds his hamstring, 2006. Credit: Action Images/Carl Recine.Livepic
Tendon injuries account for 30-50% of all injuries reported to physiotherapy clinics and with 30% of all running injuries relating to tendon overuse(1). Tendinopathies are complex in nature to manage and may result in a long lay-off from sport and long-term damage if inappropriately treated. High hamstring tendinopathies (HHT) are less commonly diagnosed than other tendons of the lower limb and may present as either deep buttock pain or posterior thigh pain(2).
The purpose of this review is to provide insight into the conservative management for HHT and when to explore injection therapy. High hamstring tendinopathies may also be referred to as proximal hamstring tendinopathy (PHT) or hamstring origin tendinopathy (HOT). The research pertaining to conservative treatment for HHT is limited, and therefore some of the research provided in this review is related to tendinopathies of other major tendons of the lower limb(2).
Anatomy of the hamstring muscle
The hamstring muscle comprises three separate muscles:
- semitendinosus,
- semimembranosus, and
- biceps femoris (long head).
They all originate at the ischial tuberosity of the pelvis (see figure 1). The short head of the biceps femoris, in contrast, originates at the posterior aspect of the femur on a ridge known as the linear aspera. The hamstring muscles course down the posterior thigh with the biceps femoris (long and short heads) attaching laterally to the head of the fibula, whereas the semitendinosus and semimembranosus attach medially to the tibia. The hamstring muscles are innervated by the tibial branch of the sciatic nerve and facilitate hip extension and knee flexion(3).
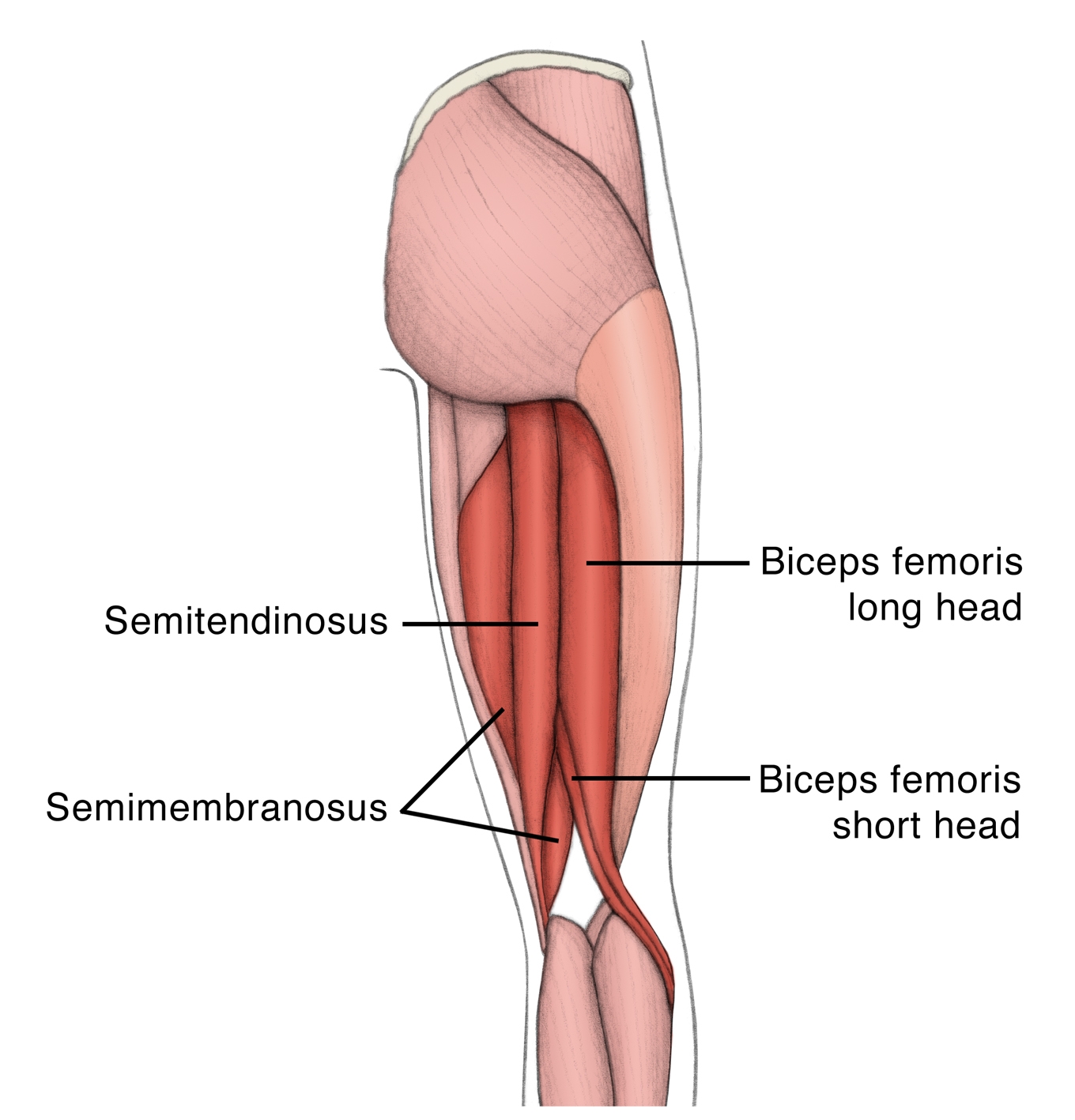
Figure 1: The hamstring muscles with the arrow indicating the primary location for pain in HHT
Pathophysiology
The term tendinosis is used to describe the process of degeneration in tendinopathy, although many still use the age-old term of tendinitis. The ‘itis’ implies an inflammatory response is present but intratendinous degeneration is defined as being hypoxic and calcific from factors such as reduced blood flow, aging and microtrauma rather than presenting as an inflammatory response(4). A hypothesis explaining the pain experienced in tendinopathy is from the ingrowth of the vessels and nerves termed as ‘neovascularisation’, which causes a pain response from the swelling of an irritable tendon(5).
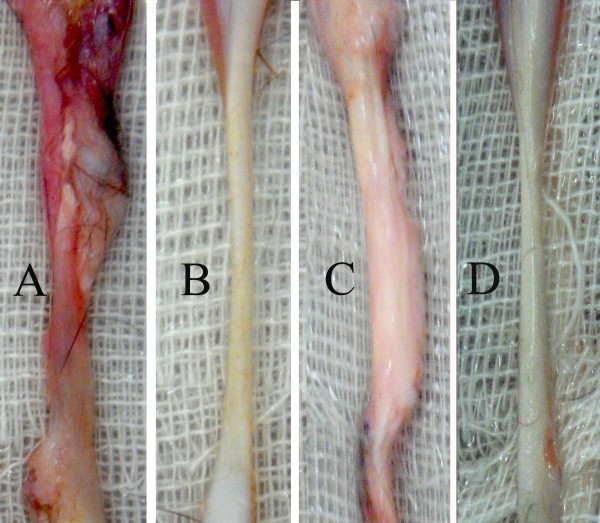
Figure 2: Pathological progression of tendinopathy
The pathological part of a tendon appears grey and dull as it loses its glistening white appearance(6). The illustration of an injured tendon in figure 2 is not tendinopathy and was a tendon removal coupled with surgical repair of the superficial digital flexor tendon in rabbits. However, the authors drew comparisons to the healing stages in tendinopathies in humans at similar timelines of 28 and 84 days (four and 12 weeks)(6). The picture illustrated in section C shows the biomechanical properties of the immature collagen fibers and is significantly lesser than the normal tendon in sections B and D.
Eccentric loading guidelines used for tendiopathies(1)
One REP MAX to determine load should be avoided to prevent further damage. Load should be determined by number of repetitions rather than weight. 3-4 sets of 6-12 repetitions to start initially. 4 sessions per week as opposed to daily. Lift with two legs and lower with involved leg.
Clinical assessment
The proximal hamstring tendon is placed under the greatest load when the knee is extended and the hip is fully flexed. Running, or running-related sports, predispose an individual to tendinopathy due to the extended duration the hamstring tendon is held in this eccentrically loaded position(7). Additional factors exposing an individual to HHT may include low hamstring-to-quadriceps muscle ratio, inadequate warm-up protocols, previous hamstring injury and reduced hamstring flexibility(2). Other factors may include pelvic dysfunction and decreased activity of the core stability muscles and therefore, a broad assessment should be undertaken with the onset of deep buttock pain(3).
Symptoms of HHT may include pain during repetitive eccentric loading, acceleration during running and in extreme cases sitting on hard surfaces. The taking-the-shoe-off test, resisted by the uninvolved foot, has been shown to have a high sensitivity and specificity of 100% for hamstring-related injuries and should be considered for use with a suspected HHT(8). Typically pain is felt on active and passive stretching of the hamstring muscles at the origin attachment, with pain also experienced on palpation of the ischial tuberosity(2). In addition, there is minimal decrease in strength of the hamstring muscles with knee flexion or hip extension in isolation and no neurological deficit is present unless further lumbosacral spine pathology is involved(2). Pain is often observed when the knee is extended with the hip fully flexed, placing the greatest load on the hamstring muscles, such as the terminal stage of the swing phase in gait.
Box 4: Rehabilitation course
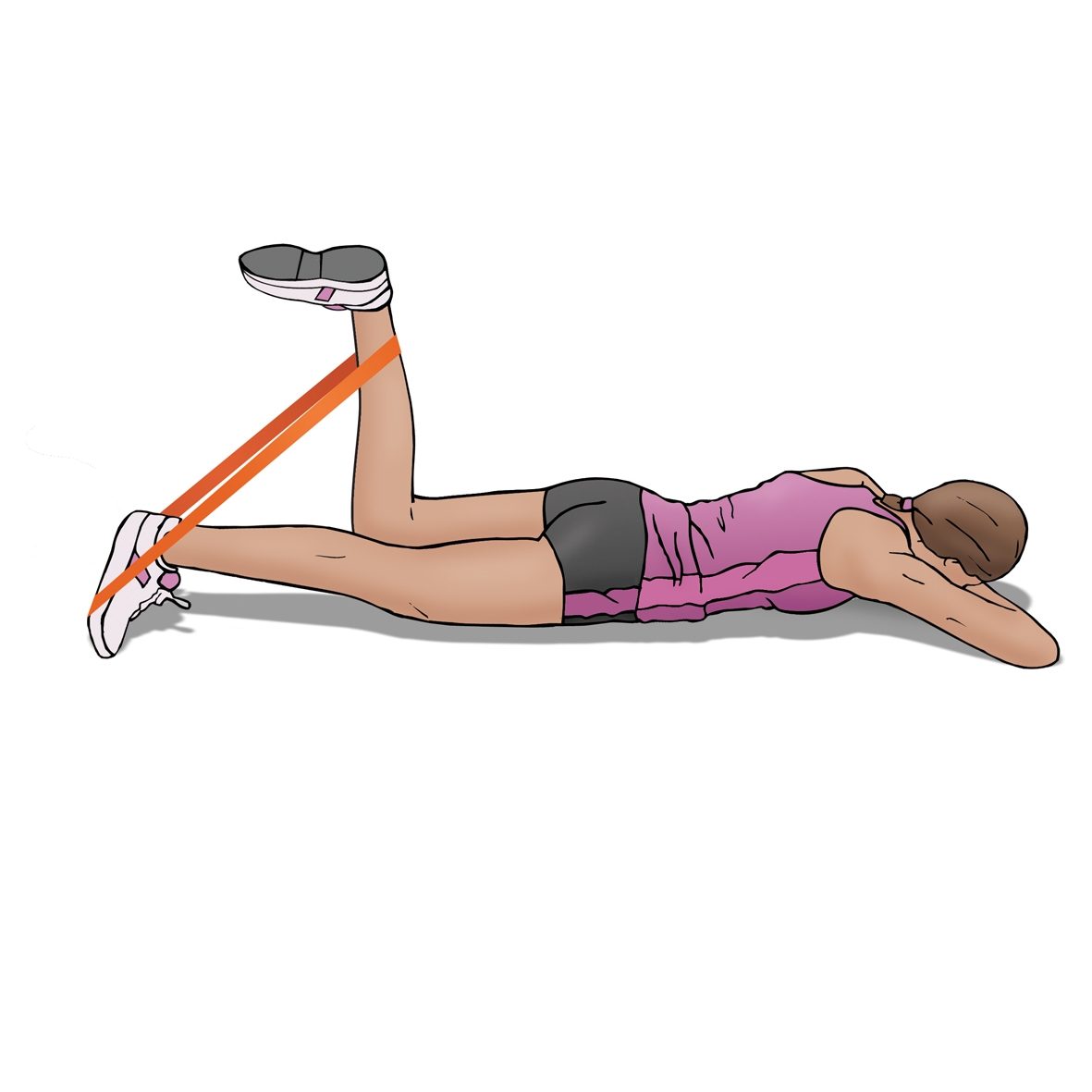
Visit 1: a) prone eccentric hamstring curls (3x10 reps, twice per day), b) supine hamstring stretch.
a) Prone hamstring strengthening
b) Supine hamstring stretch
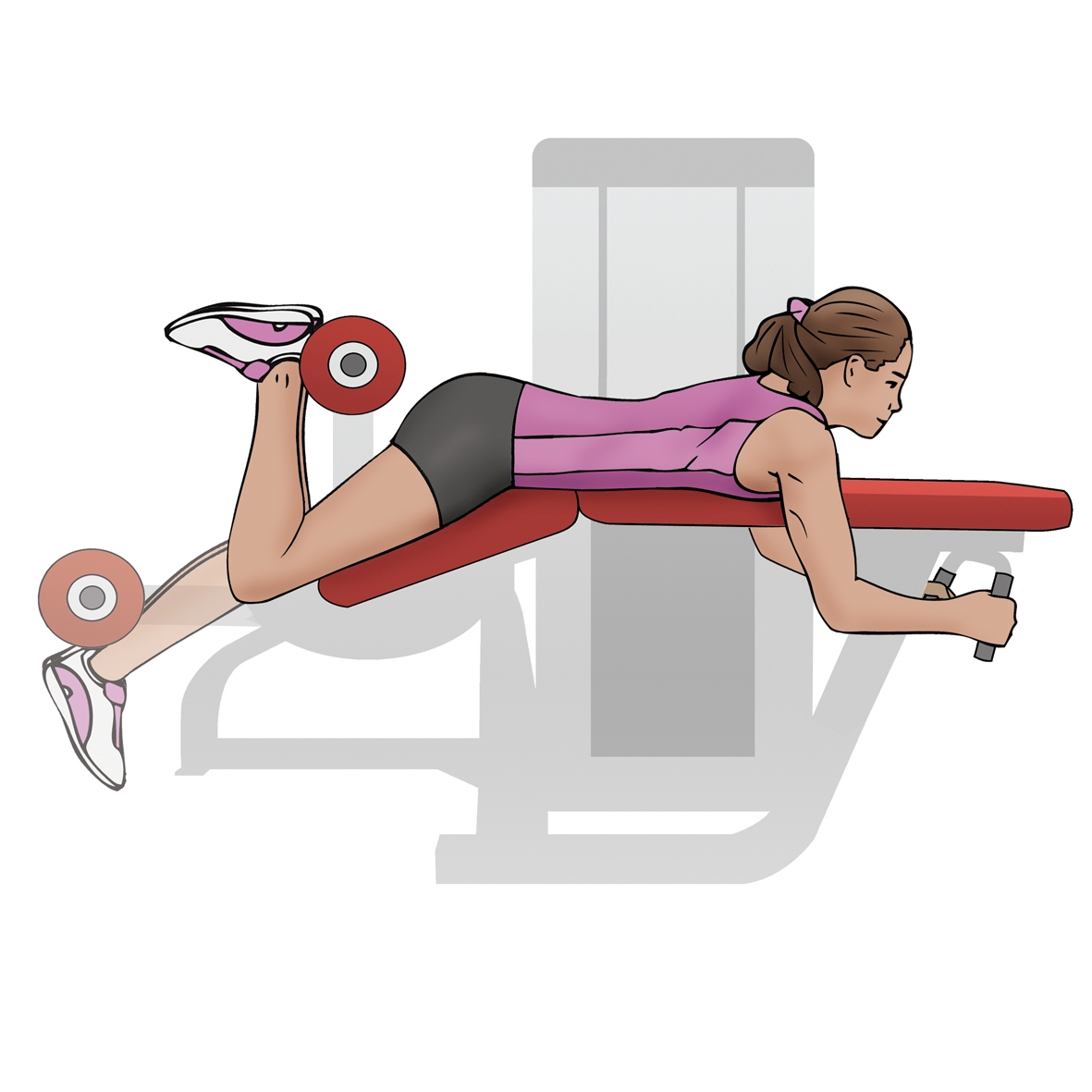
Visit 2-4: Warm-up bike, c) Prone eccentric hamstring curls using a machine (lift with two legs, lower with one), resisted hips extension, open chain quad strengthening and contract relax hamstring stretch. a)
c) Machine hamstring strengthening
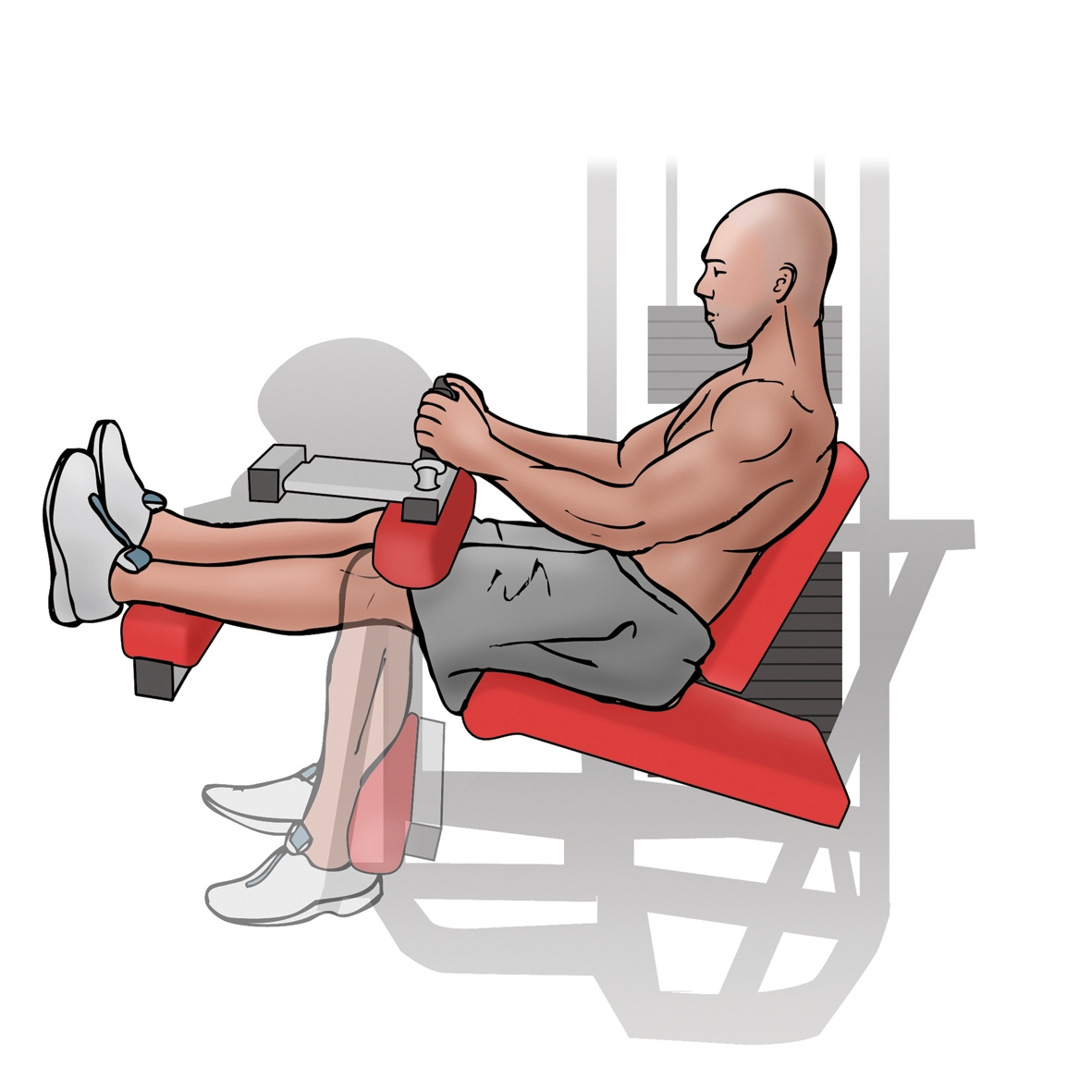
Visit 5-8: Warm up bike, prone eccentric hamstring curls, d) seated hamstring curls from 0-90 degrees.
d) Seated hamstring curls
Visit 9-12: Warm-up bike, prone eccentric hamstring curls, seated hamstring curls from 0-90 degrees, e) unilateral bridging, f) standing eccentric exercise in the form of good mornings.
e) Unilateral bridging
f) Good morning
Visit 13-16: As previously completed with the addition of g) lunges, h) single-leg squats and i) Nordic curls.
g) Lunges
h) Single-leg squats
i) Eccentric Nordic curls
Conservative treatment
An eccentric contraction, which refers to increased tension of a muscle-tendon unit during lengthening, has been widely used as a treatment tool for tendon pathologies(7). Tendons require seven and a half times less oxygen than skeletal muscle does, and during the eccentric phase oxygen consumption rarely rises to more than twice its resting value(9). Research compiled during the 1970s indicated that faster concentric contractions demand a greater oxygen supply and, as a result, an increase in heat production and cellular metabolism occur(10). Consequently, a greater amount of waste products are transported to the active site potentially causing a chemical reaction of nerve endings and an increase in pain(10). Therefore the justification for eccentric exercise in the management of tendinopathies is provided as opposed to concentric contractions.
More recent research has suggested that eccentric exercise promotes collagen fiber cross-linkage within the tendon which facilitates remodeling of the injured tissue(7). This was supported by Langaard and colleagues who found that 12 weeks of eccentric loading of the Achilles tendon increased the collagen synthesis rate of type one fibers (5). It has been stated that pain and neovascularisation are reduced following a different 12-week eccentric training program for Achilles tendinopathy(11). A period of 12 weeks is considered adequate for tendon regeneration to occur, although positive results may be observed sooner.
A case study was published in the Journal of Manual and Manipulative Therapy of a 41-year-old female recreational runner who completed five three-mile runs per week(7). The patient had an onset of right buttock pain but was able to continue training at a lower intensity for 12 months. The patient then developed left buttock pain with worsening pain on her right side which forced the patient to cease running completely. The patient’s objective was to make a full return to running. Pain ranged from 0-6/10 on the VAS scale, depending on activities. The pain was experienced on brisk walking, getting in and out of cars and sitting on hard surfaces and when taking shoes off (‘taking a shoe off test’).
An assessment of the patient in the case study indicated straight leg testing with ankle dorsiflexion and slump tests were negative bilaterally and pain-free lumbar spine movements with overpressure in all directions were also negative(7). Pain in the buttock with lumbar flexion with her knees fully extended was, however, noted. No pain on palpation of the lumbosacral spine was observed, but the patient was tender on the ischial tuberosity and the proximal two inches of the hamstring tendon. Pain was not observed on the hip quadrant test of either hip and no pain was noted on sacroiliac joint testing. Hip flexors, abductors and external rotators were all pain-free and recorded as 5/5 on the Oxford scale. Hip extension was slightly painful on both sides with a reduction in strength 4/5. Knee flexion reproduced greater pain levels on both sides with 4-/5. The pain was reproduced on both sides by passively extending the knee with the hip maintained in 90 degrees of flexion.
The treatment program of the runner presenting with HHT is highlighted in Box 4. It should be noted that an HHT won’t present in exactly the same manner, but the case study above does acknowledge other areas to be examined as part of your clinical assessment. This patient was treated with a progression of exercises over the course of 16 treatment sessions with hamstring eccentric exercises, gluteal stability exercises, stretching, and ASTYM. ASTYM is a soft tissue technique proposed to regenerate a response in the soft tissue to promote healing, but published research is yet to be provided on humans. The soft tissue technique is performed by carrying out gliding techniques in the direction of the muscle fibers. The rehabilitation exercises were selected based on the physical capabilities of the patient throughout the rehabilitation and three sets of 10 repetitions were used on each of the exercises.
The outcome of the case published by McCormack indicated a steady progression throughout with no pain on taking shoes off at visit three(7). After the eighth treatment, the patient was able to walk two and a half miles pain-free and, after 12 treatments able to jog one-mile pain free. After 16 treatment sessions, the patient reported 95% subjective improvement and was able to run for two and a half miles pain-free.
Shockwave therapy
Shockwave therapy has been cited as being an effective tool for managing HHT. A study investigated the effects of hamstring resistance training (not specifically eccentric loading) with anti-inflammatory drugs and shockwave therapy on the tendinopathy of the proximal hamstring in 40 professional athletes(12). The shockwave applied was 2500 impulses per session without anesthesia of one session per week for four weeks. The four-week study, which used a randomized controlled research design, found at three months post-study that 17 of the 20 athletes who received shockwave had a reduction in pain of 50%, whereas in the treatment group, only two patients received a reduction in their pain by 50%. Although the comparison between the shockwave versus treatment group shouldn’t be contrasted against eccentric training, it is substantial evidence for the use of shockwave in treating HHT.
Steroid injection therapy
Research on tendinopathies has indicated that 20% of patients will remain as symptomatic at three to six months after a conservative management program has been applied(13). Further interventions should be explored at this point and one of the options is in the form of injection therapy. It should be noted that once injection therapy has been carried out it is key that the conservative management program be continued for optimal results.
A fluoroscopy-guided peritendinous corticosteroid injection was provided for 18 athletes with HHT diagnosed by MRI(2). The follow-up, on average being 21 months, indicated by a questionnaire that pain had significantly reduced from 7.22 pre-injection to 3.94 post-injection. The results also indicated that athletic participation had significantly increased from 28.76% to 68.82% with 38.8% of patients being completely asymptomatic at a mean follow-up of 24.8 months. There was no documentation of a conservative program and this is the limitation of the research. Had a structured management program been provided it would be useful to draw on the results obtained and draw comparisons between the groups.
Researchers from the University of Copenhagen studied the effects of corticosteroid injections, eccentric training and heavy slow resistance training on patients with patellar tendinopathy(14). Using a randomized controlled single-blind research design, 39 male participants were assigned to one of the three intervention groups. Factors measured were function, symptoms, pain, tendon swelling and vascularisation and tendon mechanical properties prior to the study, at 12 weeks and at six months following the study.
The results of the research yielded that each intervention significantly improved pain, function, and symptoms during the first 12-week period. Furthermore, pain, function, and symptoms continued to improve in the eccentric and heavy slow resistance training groups at the six-month follow-up but had significantly reduced in the corticosteroid injection group. Also at the six-month follow-up, the heavy slow resistance training group indicated that they were the most satisfied with the treatment outcome which coincided with increased collagen turnover. Although the corticosteroid injection alone provided positive initial results at 12 weeks, the results were not substantive for the long-term clinical effects.
Summary
A rehabilitation program devised for HHT should be based on the findings from the initial examination and the load in which the hamstring muscle can withstand. Furthermore, the complexity and load of the rehabilitation exercises should be specific to the patient’s pain tolerance. If the tendon isn’t responding to conservative treatment then steroid injection is an avenue to explore depending on the patient’s age and current symptoms after a period of at least 12 weeks. To draw greater comparisons between steroid injection therapy and a conservative management program, research should endeavor to contrast the effects of both forms of treatment using randomized research design. The current review is unable to provide a firm conclusion at this point as further research is required.
You need to be logged in to continue reading.
Please register for limited access or take a 30-day risk-free trial of Sports Injury Bulletin to experience the full benefits of a subscription. TAKE A RISK-FREE TRIAL
TAKE A RISK-FREE TRIAL
Newsletter Sign Up
Subscriber Testimonials
Dr. Alexandra Fandetti-Robin, Back & Body Chiropractic
Elspeth Cowell MSCh DpodM SRCh HCPC reg
William Hunter, Nuffield Health
Newsletter Sign Up
Coaches Testimonials
Dr. Alexandra Fandetti-Robin, Back & Body Chiropractic
Elspeth Cowell MSCh DpodM SRCh HCPC reg
William Hunter, Nuffield Health
Be at the leading edge of sports injury management
Our international team of qualified experts (see above) spend hours poring over scores of technical journals and medical papers that even the most interested professionals don't have time to read.
For 17 years, we've helped hard-working physiotherapists and sports professionals like you, overwhelmed by the vast amount of new research, bring science to their treatment. Sports Injury Bulletin is the ideal resource for practitioners too busy to cull through all the monthly journals to find meaningful and applicable studies.
*includes 3 coaching manuals
Get Inspired
All the latest techniques and approaches
Sports Injury Bulletin brings together a worldwide panel of experts – including physiotherapists, doctors, researchers and sports scientists. Together we deliver everything you need to help your clients avoid – or recover as quickly as possible from – injuries.
We strip away the scientific jargon and deliver you easy-to-follow training exercises, nutrition tips, psychological strategies and recovery programmes and exercises in plain English.