Proximal Hamstring Tendinopathy: A challenge for athletes and clinicians

Tendon injuries are a common sporting problem. Whilst much attention is focused on the more common issues of Achilles and patella tendon pain, proximal hamstring tendinopathy (PHT) is a condition that, if inappropriately managed, can lead to persistent performance decrements in the first instance and thereafter extended time loss. Sports such as the various football codes played around the world, provide the perfect combination of injurious mechanisms, with relatively high running distances and velocities, alongside a kicking technique which can lead to a long ‘shank arm’ adding further load to the region.
Anatomy
The hamstrings are a biarticular muscle group consisting of the bicep femoris (BF), semimembranosus (SM) and semitendinosus (ST). The ST and the long head of BF unite to form a conjoined tendon before attaching to the posteromedial portion of the ischial tuberosity. The SM is considered to have a deeper attachment, originating just anterolaterally to the conjoined attachment on the ischial tuberosity.It is important to note that the proximal tendon has an intimate relationship with the inferior gluteal nerve, artery, and the sciatic nerve, this often means that PHT is accompanied by a neural irritation. An appreciation of the local structures is required to assist clinicians in making differential diagnosis.
The hamstring muscles work isometrically to maintain standing posture, resisting excessive postural forward lean and excessive lumbar lordosis. When we consider the lever arm of the hamstrings in relation to the hip joint, it becomes significantly larger or smaller as the hip joint changes position. Consequently, the hamstrings will be required to work in situations where there is forward flexion in the hip or trunk, with the proximal hamstring tendon and its attachment to the ischial tuberosity subjected to higher loads when placed in this position(1). In running, during terminal swing phase, the hamstrings are required to rapidly decelerate knee extension. Energy storage in the late-swing/early-stance stretch-shorten cycle is thought to be a potential risk factor for PHT.
Aetiology
Cook and Purdam presented a model describing the progression of a tendon from normal to pathological(2). In this model, a tendon can adversely react to imposed load by increasing tenocyte activity and protein synthesis. At this stage with appropriate load modification, the tendon heals well. However, should load exceed tolerance, the tendon’s collagen structural framework can be disrupted.Whilst there is limited research on PHT, it is thought that the pathological presentation is similar to findings in the other weight-bearing tendinopathies(3). From the limited studies available, PHT presents with localised tendon pathology at the enthesis, bone oedema and insertional tendon clefts(3-5).
In the athletic population tendinopathy represents a maladaptive response to an increase in load. The extrinsic risk factor of a causative spike in training load over days or weeks, coupled with intrinsic biomechanical and movement pattern dysfunctions, leads to deterioration in the tendon health. Other extrinsic risk factors that require consideration include:
- An injury to another tissue or structure above or below the chain
- A change in footwear
- A change in terrain
- A change in external resistance
The clinician is also encouraged to review the intrinsic risk factors related to tendinopathy. It is thought intrinsic risk factors reduce the threshold for tendon pain and pathology. These include; age, body mass index, metabolic issues, hormonal change, and fluoroquinolone antibiotic use (see Sports Injury Bulletin issue 153 for an in-depth discussion of fluoroquinolones in sport).
Subjective assessment
Often the athlete will report a gradual insidious onset of localised deep buttock pain and tightness over the ischial tuberosity and gluteal fold region. Running at higher velocities is often implicated as an aggravating factor, but like many tendinopathies it will often ease following a thorough warm-up.As the condition deteriorates, the pain fails to settle, limiting top end pace and worsens with increased duration of running volume or repeated provocative movements. A thorough history may reveal the positions of provocation place the proximal hamstring tendon under compressive load at the ischial tuberosity, such as the dead lift, squat, lunge, high step up and repeated kicking out of hand. Moreover, periods of prolonged sitting increase compression and can aggravate symptoms. As such, it is not just training loads that need to be fully understood but also sitting loads. It is unsurprising therefore that this condition is often seen in athletes that are also studying or have high driving mileage.
Whilst completing the subjective assessment it is important to listen to the way the athlete describes their pain and their relationship to pain. An understanding of the psychosocial influencers of pain in PHT will dramatically improve a clinician’s chances of engagement if detrimental beliefs are held by an athlete.
Fear avoidance, anxiety and a heightened attention to PHT may lead to amplification of neural signalling within the central nervous system that then elicits pain hypersensitivity(6,7). This effect has been found in other tendinopathies(8), and if not addressed can dramatically affect recovery. The reader is encouraged to read the work of Woolf, addressing central sensitisation(7).
Differential diagnosis
It has been observed that PHT cases in athletes are often associated with a past medical history of hamstring strains and/or sciatic nerve compromise. It is not uncommon in more chronic cases for supposed isolated hamstring strains to coexist(3). Once a detailed subjective history has been established, the clinician must consider the other structures that may present in an analogous way. Utilising a systematic approach to inclusion/exclusion will ensure all appropriate co-morbidities and differential diagnoses have been accounted for (see table 1).| Possible differential diagnosis | Exclusion criteria |
|---|---|
| Lumbar spine facet arthropathy,Disc degeneration,Radiculopathy | No diffuse leg referral+ve hamstring load testsLumbar palpation (NAD) & -ve quadrant test |
| Hip joint ischiofemoral impingement+/- Quadratus femoris abnormalities | Femoral external rotation in hip neutral -veMRI -ve (No loss of space and QF normal)Flexion-adduction-internal rotation (FADDIR) -ve |
| SIJ somatic referral | Lasletts’s SIJ provocation tests -ve |
| Sciatic nerve compression | Sciatic tenderness at QF -veSlump test +ve hamstring but no change with sensitisers (Hip adduction/internal rotation)Modified slump (lx extension) Differential test for specific comparison to PHT*Coexisting pathology possible |
| Piriformis syndrome | Sciatic nerve non-tender at piriformisNo further provocation with piriformis stretch/contraction or slump with Add/IRMRI imaging -ve |
| Gluteal tendinopathy | +ve Hamstring load tests-ve gluteal load testsMRI -ve |
| Ischio-gluteal bursitis | Pain with stretch and localised palpationIrritable symptoms with sittingMRI and ultrasound -ve |
| Partial or complete tear of the gluteal or hamstring muscle/tendon | Gluteal and hamstring tests +ve but MRI and U/S findings -ve for muscle or tendon tear respectively |
| Partial or complete tear of the gluteal or hamstring muscle/tendon | Tenderness over ischial ramusMRI imaging -veHigh suspicion in female triad athletes |
| Adductor magnus pathology, tear or tendinopathy | Adductor tests -ve,PSST adductor stretch and resist -veMRI -ve |
| Intrinsic risk profile:Gender, systemic inflammatory disease, age, BMI, hypercholesterolaemia, diabetes, hormonal disturbance and recent fluroquinolone-based medication | -ve blood testsBMI within normal limitsNon perimenopausal with normal menstrual cycleNo antibiotics within last 12/12 |
*Adapted from Caine 2017(9)
Imaging
Most often, the diagnosis of PHT can be made without radiographic imaging, but it can be helpful to rule out other structures that may be implicated in the dysfunction. magnetic resonance imaging (MRI) is the gold-standard technique and PHT commonly presents with thickening and surrounding oedema, along with changes mimicking bone marrow oedema, which show this is more an isolated tendon issue.However, what is worthwhile noting are the findings of a recent study showing these same signal changes are found in asymptomatic patients just as readily as symptomatic cases(4). The importance of treating the individual based on the clinical presentation and not just the reported findings must be at the forefront of a clinicians mind - ie ‘treat the man not the scan’!
Objective
Whilst validated tests for PHT with high sensitivity and specificity are limited, three passive stretch tests (bent-knee stretch, modified bent-knee stretch and the Puranen-Orava test) have moderate to high validity and high sensitivity(10). A test is positive when proximal hamstring pain is elicited with compression on the ischial tuberosity. However, these tests have been found to be less sensitive for early/less symptomatic PHT presentations.The author uses a straight-leg variant ‘raging bull test’ in a hip flexed position. The athlete is asked to isometrically pull their heel down and back into the floor for a few seconds; pain provocation during or immediately on release can indicate PHT. The pain provocation test continuum looks to place the tendon under progressive compression and tensile load by increasing hip flexion angles. Tests are initially completed slowly, tempo is accelerated if asymptomatic at slower pace. The pain score should increase with load across these tests (see figure 1).
Figure 1: PHT pain provocation tests

A: Short level bridge (low-load test) - upper left
B: Long lever bridge (moderate load test) - bottom left
C: ‘Raging bull’ pull variant (moderate-high load test) - upper right
D: Arabesque (high load) - middle right
E: Single leg dead lift (high external load test) (Cook, 2014). - bottom right
PHT has the potential to present with hamstring muscle atrophy and weakness. The use of a dynamometer to assess hamstring strength through both knee flexion and hip extension is certainly a good method to measure deficiencies(11,12). Other more accessible capacity tests are the single leg bridge to fatigue test(13)and a prone hamstring curl, with a degree of hip flexion to mimic the position of running(14). This achieves good activity within bicep femoris and semitendinosus whilst minimising gluteus maximus action.
Once rudimentary hamstring strength has been established, it’s important to assess the kinetic chain for weakness and/or imbalances. These may be part of the underlying movement dysfunctions that contribute to the development of PHT during periods of increased training load. Gluteus maximus (GMx) and medius (GMd) weakness are thought to expose the proximal tendon to further load by an increase in hip adduction and/or contralateral pelvic drop during squatting or lunging(15,16).
Grimaldi’s active lag test although targeted at GMd gives a good clinical indication of weakness, especially when findings are compared to singe leg squat control, which may highlight excessive anterior tilt and hip flexion(17). The increased anterior tilt increases hamstring stretch during functional positions; therefore working towards pelvic neutral has the potential to reduce proximal hamstring load.
The importance of adductor magnus assessment is highlighted by its intimate attachment with semitendinosus(18). Due to their shared attachment, it is not uncommon to find adductor tendon changes - as well as PHT - on imaging. If an athlete has an adductor dominance compared to abductor, this has the potential to exacerbate ischial shear. The author believes the adductor/abductor ratio should form part of a more detailed assessment.
Running analysis
Running analysis is an important assessment in PHT management. Running techniques that are characterised by over-striding, excessive forward trunk lean, increased anterior pelvic tilt, pelvic instability and poor hip extension can increase compressive loads at the ischial tuberosity. Research has demonstrated increasing cadence reduces stride length and hip flexion at foot strike(19)and increases gluteal activity in terminal swing(20). These changes have the potential to reduce proximal hamstring tendon compression at the ischial tuberosity.Load management
In-season management of PHT requires careful load management, and the ability to reduce compressive loads from weights programming and training sessions can lead to an optimised output from a pathological tendon. This may also include the removal of dead lift work, reduced plyometrics work and kicking volume. The use of a visual analogue score (VAS) can be helpful to track an athlete’s symptoms. A VAS of 0-3 out of 10 that settles within 24 hours demonstrates tolerance to an appropriate load. However, symptoms lasting longer than 24 hours are indicative of deleterious loading and further removal of provocative activities is required.The replacement of running volume with alternative conditioning requires careful consideration. Standing cycling, swimming and deep water running provide useful options. However, in some cases this level of offload is unhelpful as it can create an issue somewhere else or lead to a unforeseen deleterious spike when the athlete returns to training. With this in mind, and prior to seeking alternatives, can we constrain running in any other way to allow optimal tissue load whilst PHT continues to resolve?
Options may include running mechanics and reduced pace ‘straight-line’ conditioning runs, with the primary focus instead being biomechanical correction. Athlete education is therefore vital to promote compliance throughout the process, and avoidance of positions of provocation needs to include all activities. In addition, limiting sitting duration and use of offload cushions will help alleviate ischial compression and the associated pain.
Rehabilitation
The progressive load management of PHT requires careful monitoring. A good clinical assessment tool to assist with this is the VISA-H questionnaire(21). This allows clinicians to track progress; however caution should be applied to the frequency of use as obsessive tendencies in a PHT sufferer can lead to amplified pain perception.At present there is no consensus in the literature regarding the best management pathway for PHT (see table 2). Positive results have been reported in case series/studies for conservative management(15,22-24). Goom and Malliaras also provide a staged approach to rehabilitation of PHT(25). The author will reinforce this pathway and build upon its recommendations once an athlete has returned to competition.
Each case of PHT will have different intrinsic and extrinsic factors contributing to the pathological change. It is important to address these factors throughout the rehabilitation process. Progression of load and sport-specific involvement should be based on pain provocation monitoring. A prescribed time frame is ineffective; rather, the careful monitoring and review of response to load is currently the best strategy to return an athlete to competition in a safe and timely fashion.
Monitoring of pain at the same time each day with a load provocation test is vital during rehabilitation. The pain provocation test chosen will be specific to each case, commonly used tests are presented in figure 1. It is important to counsel the athlete that a 0-3/10 pain is acceptable during warm-up but is expected to dissipate and this may return toward the end of, or post session. Reinforce to the athlete that this is a positive adaptation. However, bear in mind that we as clinicians want to see this post-loading response settle within the next 24 hours(26).
Table 2: Examples of phase-one exercises and possible progressions
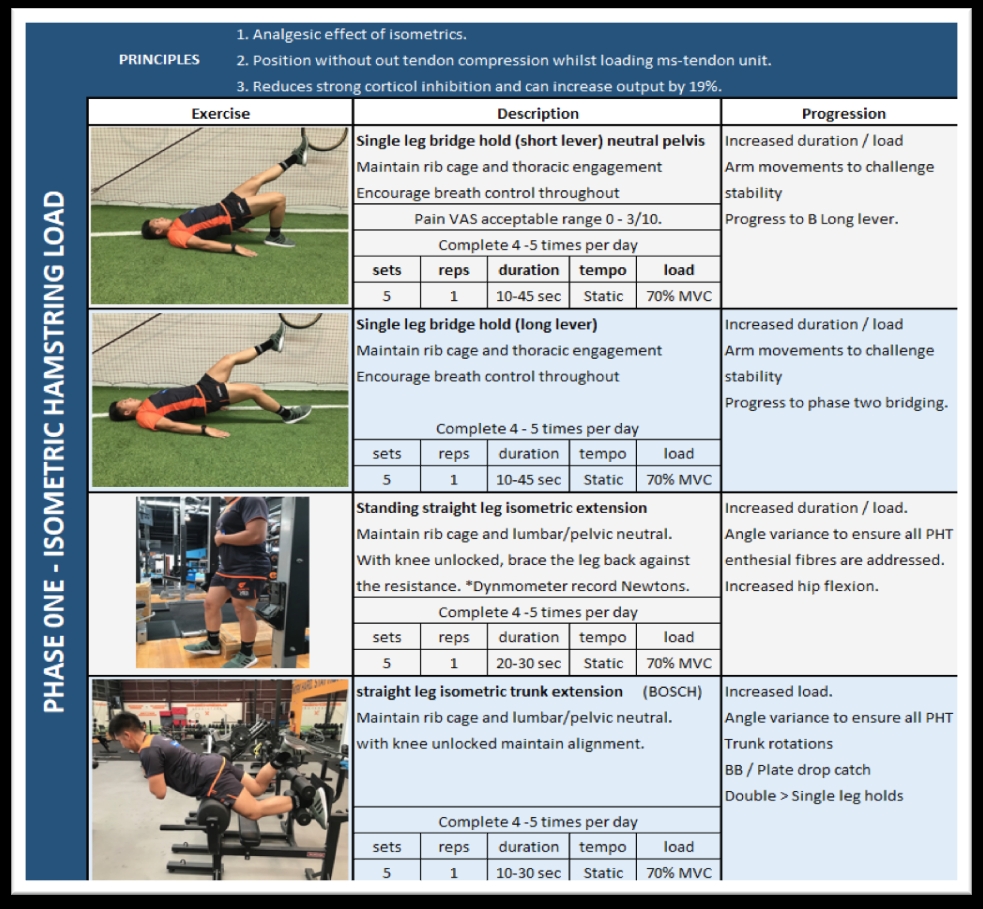
Phase one: Isometric loading
The primary focus of phase one is to introduce load without compression of the tendon. Isometric exercise has been shown to have an analgesic effect for tendinopathy(27). Five sets of 45-second holds at 70% maximal voluntary contraction have been shown to reduce patellar tendon pain for at least 45 minutes, and provided longer relief than isotonic exercises(8). In practice, the duration of the hold will be dictated by the degree of pain provocation; an entry level may be 10 seconds.A way to monitor (and help increase) the relative force applied is to use a dynamometer or a pressure cuff to measure the mmHg applied whilst a single leg bridge is performed. Cook and Purdam [28], advocate repeating the exercise several times per day(28). Isometrics provide a useful prognostic indicator; if symptom reduction occurs after completion of the exercise, it may be appropriate to trial either an increased isometric force or duration.
Phase two: Strength isotonics
Progression to phase two will be guided by the introduction of a degree of hip flexion. When we look to progress load, it is important to allow adequate time for tissue remodelling by respecting a 2-3-day response to new load. By avoiding two loading spikes in three days, we allow appropriate time for physiological adaptation and central processing.Phase 2 targets a return to strength and capacity in progressive functional ranges (see table 3)(29). Eccentric exercises and heavy slow resistance loading have both been identified as having beneficial effects in tendinopathies other than PHT(30). Heavy slow resistance (HSR) has been found to have improved compliance and achieve greater collagen turnover than submaximal eccentric loading(31).
Table 3: Single leg bridge progressions, prone leg curl, Nordic hamstring, high box bridges* double leg up single leg hold. Lx/pelvic challenge with shared load.
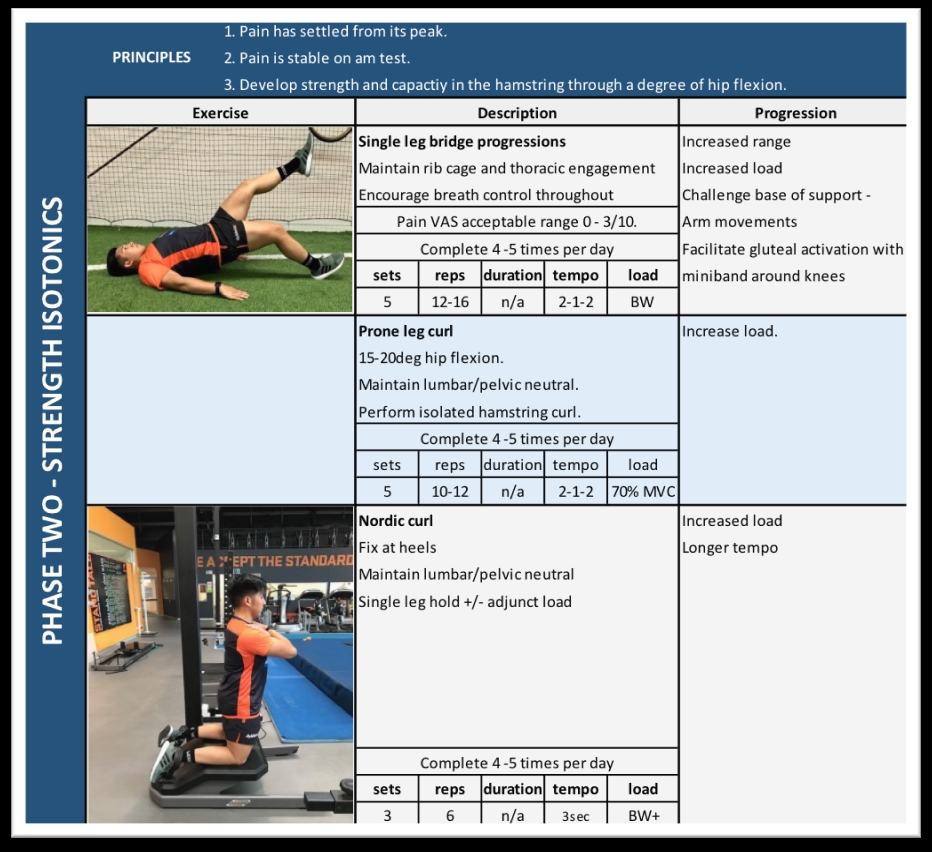
The relative bias of the working muscles during movements needs to be appreciated when loading; the knee dominant movement of a leg curl biases the bicep femoris, whilst a hip dominant movement such as dead lift or kettle bell swings targets semitendinosus. When considering rehabilitation planning, the graded exposure to specific load must consider knee or hip dominance and the muscle group targeted. This can then be used as a progression of optimal load.
Phase 3: Isotonics plus hip flexion
Phase 3 progression involves increased degrees of hip flexion (70-90 degrees). The addition of loaded hip flexion has the potential to cause flare ups as the compressive force of PHT is increased. Therefore, the exercises must respect a high, low, medium day loading plan. An example pre-season program (relative to PHT reload) is as follows:- Monday (high) – Prone curl and Nordics incorporated into lower limb weights (note. Running requirements to be completed prior to weights).
- Tuesday (low) – Basic isometrics incorporated in upper limb weights.
- Wednesday (medium) Body weight circuit and single-leg bridge progressions plus mechanics.
- Thursday – Recovery.
- The load cycle can then be repeated.
The order can be manipulated throughout the season as the demands change. The appropriate exercises for this phase include hip thrusts, step ups, walking lunge, deadlifts and Romanian deadlifts. A loaded step up is an excellent posterior chain challenge with GMx, GMd and BF recruited(32), while lunges have been shown to recruit the proximal BF as well as AM(33). The author recommends a high-box bridge progression with weighted perturbations from various angles to challenge the tendon attachment at the ischial tuberosity from different force vectors (see figures 1 and 2).
Figures 1 and 2: High box single leg bridge with med ball throw catch perturbations


Angle variance to challenge hamstring and adductor magnus interface whilst challenging gluteal/lumbar/pelvic control.
Phase 4: Energy storage
Phase 4 is vital in the sporting population with the importance of energy storage and loading impacts. Reintroduction of power should be progressed when appropriate bilateral hamstring strength and lumbar/pelvic control has been identified. As the energy storage capacity requires control to mitigate any high compressive forces, it is recommended to start with reduced hip-flexion angles and progress as symptom provocation allows.Exercise examples will include; basic skips, sled pull, prowler push, split squats, and kettle bell swings. The athlete should already be back to steady state running at this stage. The goals being; running mechanics re-education and building a volume base in anticipation of future training load. A final consideration is the progression to sport-specific activities such as kicking. This requires attention due to the large compressive force potential - therefore intensity and volume are important considerations at this stage.
Phase 5: Elastic function and return to competition
Multidirectional sports require progression in all planes of movement - lateral, rotational and step/cut. The enthesis must undergo appropriate load in differing angles to allow optimal remodelling for sport-specific demands. The physiotherapists, conditioning coaches, coaches and player must all work together to continue the control and efficiency of movements. As the volume of training increases, the exercise requirements will need to reduce otherwise this will lead to a spike in tissue loading.Progression from phase 4 may include an increase in dynamic movement; explosive skips, bounding, fast prowler push, sled drag sprints, alternate leg split squats. Advancing from the Nordic curl to the razor curl increases the load to the proximal tendon in elite athletes, and it also more closely mimics the loaded hip/knee joint angulation and contractile variation found in sprint mechanics(34). This high-demand exercise will require robust lumbar/pelvic control whilst challenging the hamstring musculotendinous interface(35). The razor curl may be too high a load exercise for athletes who do not exhibit kinetic control, but adaptations can be made to achieve this (see figure 3).
Figure 3: Razor curl +/- slide board assist


Ongoing management
In the author’s opinion, on-going management of any athlete with tendinopathic history is vital. In team sports, careful load monitoring via GPS provides accurate on-field review, whilst the strength and conditioning and physiotherapy team play a vital role in review and management of the acute-to-chronic exercise load and its relationship with the on-field load.Load management strategies should be used to anticipate these relative high-risk periods. Isometrics should form part of an individualised activation plan conducted prior to sessions. The analgesic effect can last for 45 minutes and can improve output by approximately 20%, allowing the athlete to go into the session feeling prepared. Session structure should remove ‘dead’ time as standing around often leads to symptom aggravation and can lead to adoption of sub-optimal postures predisposing PH tendon compression load.
Manual therapy and other interventions
The load capacity of the tissue is the key to resolving PHT. Passive modalities are unlikely to implicitly affect this. However, interventions that may optimise tissue function by addressing restrictions and tonal changes may facilitate improved movement patterns when the tissue is loaded(36). Dry-needling and soft tissue techniques have been advocated in some case series/case reviews, but evidence for their benefit is limited (15,24). Extracorporeal shockwave therapy has been shown to offer good pain modulation in more chronic (non-reactive) cases, however further research is required. Platelet-rich plasma injections are another modality utilised in a bid to stimulate healing but again there is insufficient and conflicting evidence for their use(37).Conclusion
PHT is a challenging condition that requires careful differential diagnosis. It is important to gain an in-depth history from the athlete to understand the mismatch between tendon capacity and load. PHT requires appropriate education regarding pain perception to ensure athlete expectations are aligned with a progressive rehabilitation strategy. Focus must be on regaining strength throughout the kinetic chain. If symptoms are respected via careful monitoring of the response to load, this approach will allow a timely return to competition. Once the athlete has returned to sport it is our duty to ensure the athlete is empowered with the strategies to manage their condition throughout the peaks and troughs of a competitive season.References
- Running: Biomechanics and exercise physiology applied in practice. Elsevier. 2005.
- Br J Sports Med. 2016;50:1187-1191.
- Ligaments Tendons J. 2015;5:23-28.
- AJR Am J Roentgenol. 2012;198:418-422.
- J Ortho Traumatol. 2013;14:83-89.
- Br J Sports Med. 2013;47:1120-1126.
- 2011;152:S2-S15.
- Sports Med. 2014;44:9-23.
- Sports Physio. 2017;2:20-24.
- Br J Sports Med. 2012;46:883-887.
- Scand J Med Sci Sports. 2010;20:493-501.
- J Sci Med Sport. 2012;15:444-450.
- Br J Sports Med. 2014;48:675-676.
- Phys Ther. 2006;86:683-697.
- J Chiropr Med. 2011:10:93-99.
- J Orthop Sports Phys Ther. 2014;44:198-205.
- J Bodyw Mov Ther. 2010;14:294-298.
- Knee Surg Sports Traumatol Arthrosc. 2015;23:2554-2561.
- Med Sci Sports Exerc. 2011;43:296-302.
- Gait Posture. 2012;36:231-235.
- Br J Sports Med. 2014;48:448-452.
- J Orthop Sports Phys Ther. 2015;45:557-562.
- Phys Sportmed. 2005;33:32-43.
- J Man Manip Ther. 2012;20:142-146.
- J Orth Sports Phys Ther. 2016;46:483-493.
- Am J Sports Med. 2007;35:897-906.
- J Pain. 2012;13:1139-1150.
- Br J Sports Med. 2009;43:409-416.
- Br J Sports Med. 2013;47:536-544.
- Am J Sports Med. 2015;43:1704-1711.
- Scand J Med Sci in Sports. 2009;19(6):790-802(13)
- J strength Cond Res. 2012;26:3398-3405.
- J Strength Cond Res. 2013;27:3426-3435.
- J Strength & Cond Res. 2009;23:401-405.
- Sports Med. 2013;43:1207-1215.
- Br J Sports Med. 2001;35:65-69.
- Cochrane Database Syst Rev. 2014;4.
You need to be logged in to continue reading.
Please register for limited access or take a 30-day risk-free trial of Sports Injury Bulletin to experience the full benefits of a subscription. TAKE A RISK-FREE TRIAL
TAKE A RISK-FREE TRIAL
Newsletter Sign Up
Subscriber Testimonials
Dr. Alexandra Fandetti-Robin, Back & Body Chiropractic
Elspeth Cowell MSCh DpodM SRCh HCPC reg
William Hunter, Nuffield Health
Newsletter Sign Up
Coaches Testimonials
Dr. Alexandra Fandetti-Robin, Back & Body Chiropractic
Elspeth Cowell MSCh DpodM SRCh HCPC reg
William Hunter, Nuffield Health
Be at the leading edge of sports injury management
Our international team of qualified experts (see above) spend hours poring over scores of technical journals and medical papers that even the most interested professionals don't have time to read.
For 17 years, we've helped hard-working physiotherapists and sports professionals like you, overwhelmed by the vast amount of new research, bring science to their treatment. Sports Injury Bulletin is the ideal resource for practitioners too busy to cull through all the monthly journals to find meaningful and applicable studies.
*includes 3 coaching manuals
Get Inspired
All the latest techniques and approaches
Sports Injury Bulletin brings together a worldwide panel of experts – including physiotherapists, doctors, researchers and sports scientists. Together we deliver everything you need to help your clients avoid – or recover as quickly as possible from – injuries.
We strip away the scientific jargon and deliver you easy-to-follow training exercises, nutrition tips, psychological strategies and recovery programmes and exercises in plain English.









